Chemical reactions gone wrong, I’m sure we all have experienced this issue, I know I have. You add your reagents in the proper amounts with a suitable solvent and perform your reaction only to find your by-product yield was greater than your product; by a lot. So, what do you do to isolate what little product you created with maximum yield and purity without breaking the proverbial bank on a big flash column and the solvent required for the purification?
In this post I will talk about this exact situation I experienced firsthand. The reaction is one I had performed previously but with a different solvent, DCM, and at larger scale. The reactants were hippuric acid (1 gram) and α-methylbenzylamine (0.7 gram) in 4 mL of DCM. The reaction was performed at 150 °C for 15 min by a Biotage® Initiator+, Figure 1.
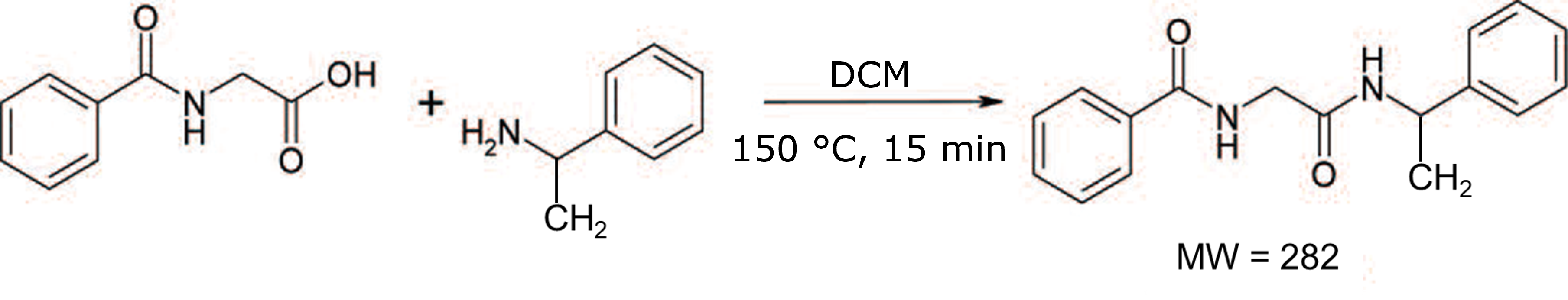
Figure 1. Hippuric acid reaction with α-methylbenzylamine.
The reaction generated a bright yellow solution which was analyzed by TLC. From previous experience, I knew the product was quite polar and that a mobile phase of hexane (60%) with (3:1) EtOAc/IPA (40%) should provide a suitable TLC separation and, therefore, purification.
To my chagrin, the TLC revealed two visible (by UV) early eluting compounds followed by a long streak of other UV absorbing compounds including my desired compound, Figure 2. I knew I had created the desired amide from the mass analysis I performed, which, of course, also showed unreacted hippuric acid and some other masses.

Figure 2. Reaction mixture TLC performed in 60% hexane/ 40% (3:1) EtOAc/IPA. The desired product was in the long streak.
So, how should I approach this crude mixture’s purification? I could use a big, 200 gram flash column and load all 1.7 grams of reaction mixture (close to the 1% load rule) consuming a minimum of 4 Liters of solvent without any success 'guarantee', or I could re-synthesize, also with no guarantee of a successful outcome.
Instead, I chose to take a greener, more pragmatic approach where the entire reaction mixture is cleaned-up with a small (10-gram) silica column followed by re-purification of the recovered product by a small (12-gram) reversed-phase column. Sure, the purification on the small column (Biotage® Sfär HC, 10 gram) would certainly be compromised by the high load (17% by weight), but I surmised that I could get some product isolated and maximize yield if I dry-loaded the reaction mix and used the TLC-based gradient.
In preparation for this high load purification, I dried the crude (mixed with 5 grams of silica) creating my dry load (Biotage® V-10 Touch). The resulting purification generated the chromatogram in Figure 3 where I determined that fraction 4 (at ~100 mL) contained my desired product.
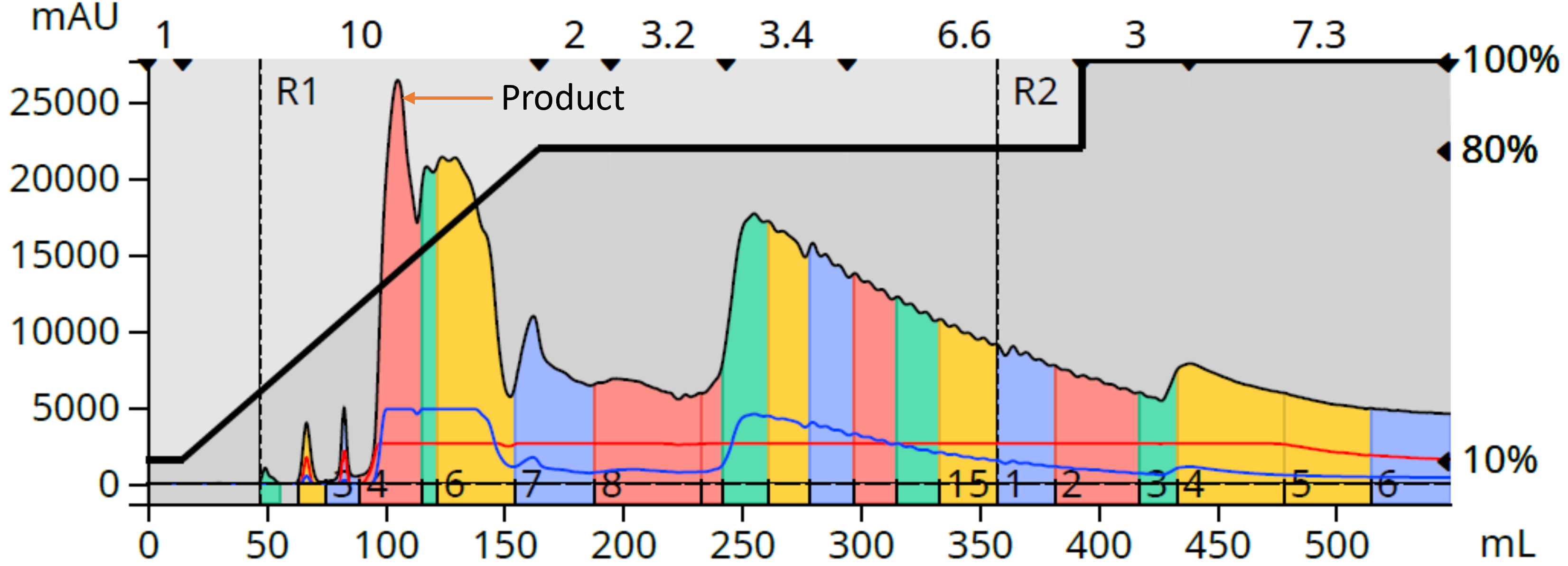
Figure 3. Normal-phase flash purification with a 10-gram silica column partially purified the desired product from a 1.7 gram crude reaction mixture load.
I evaporated fraction 4 and found it contained 0.244 grams of "product" (14.2% yield). Because the separation from the other by-products/starting materials was incomplete, my product fraction was surely impure and required re-purification. For this purification step I decided to switch to reversed-phase flash thereby using a technique called orthogonal flash chromatography, which I have seen dramatically improve product purity with other challenging reaction mixtures and natural product extracts.
So, I dissolved the 244 mg of contaminated product in 2 mL DMSO and re-purified it using a 12-gram Sfär C18 column. This generated the purification seen in Figure 4, which yielded 0.208 grams of a bright white crystalline product and a final purified yield of 12%.
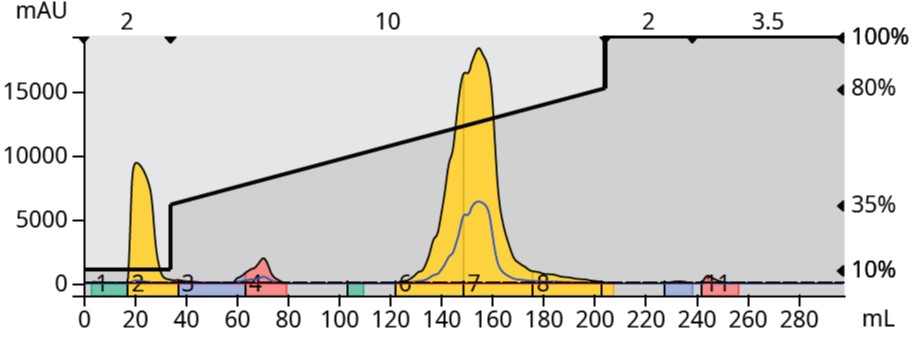
Figure 4. Reversed-phase chromatography of the normal-phase purified product generated a complete synthetic target separation and isolation.
Did this approach make sense? I say yes because the 10-gram silica column consumed only 550 mL of solvent (this much due to the gradient extending to elute the polar compounds). The "predicted" 200-gram column using the same method, including the gradient extension which would definitely occur, would have consumed 11.4 Liters of solvent! So, by downsizing my silica column and overloading it, I saved 10.8 Liters of solvent. Remember, based on the TLC, there was no guarantee the larger column would provide 100% pure product.
Also, the orthogonal reversed-phase purification used only 272 mL for a total solvent consumption of under 825 mL, a savings of 10.5-Liters; and I got my product purified.
Want to learn more about greener flash chromatography? Click the link below:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership