Synthetic organic chemistry is the genesis of new pharmaceutical and commercial chemical products. In short, it is based on the idea that two or more carbon-based compounds can be forced to react using heat, or other energy source, to create a new, novel product – but this we already know.
What are the steps most chemists follow to do this successfully?
- -Scour published literature for similar compound synthesis
- -Determine the number of synthetic steps required to create your desired compound and likely synthetic yields with each step
- -Determine the amount of each reagent needed for each reaction step
- -Determine needed equipment to perform the synthesis, post-synthesis work-up, purification, and evaporation
Once you have your raw materials, equipment, and synthetic pathway, your creative journey can begin.
- -Synthesis
- -Reaction monitoring with LC/MS, TLC
- -Quench
- -Liquid-Liquid extraction (typically aqueous extraction)
- -Scavenge water with sodium (or magnesium) sulfate
- -Evaporate to solid or oil
- -Re-dissolve for purification
- -Purify with chromatography
- -Isolate purified compound from purification solvent
- -Run next reaction step
Synthesis is performed using any number ways – round bottom flasks and a water or oil bath, scintillation vials on a hot plate, or microwave reactor, Figure 1.

Figure 1. Microwave used for organic synthesis (Biotage Initiator+)
Since most reactions create not only the desired compound but also by-products, work-up/purification is usually required. Normally, extraction of the reaction mixture to remove either the product or impurities is performed, followed by evaporation and finally, actual purification.

Figure 2. Typical work-up products include cartridge based extraction and drying tools (Biotage phase separators and drying cartridges) and a high-speed evaporator (Biotage V-10 Touch).
Since the extraction process is rarely specific for a single compound further purification is needed. This step typically involves flash chromatography, a process which can be manual or automated and includes a column filled with a solid media and solvents to elute the different reaction mixture components at different rates. Today’s synthesis labs use automated purification equipment which includes not only the column and solvents but an ultra-violet (UV) detector to separate, detect, and isolate the reaction mixture’s compounds, even complex reaction mixtures, Figure 3.
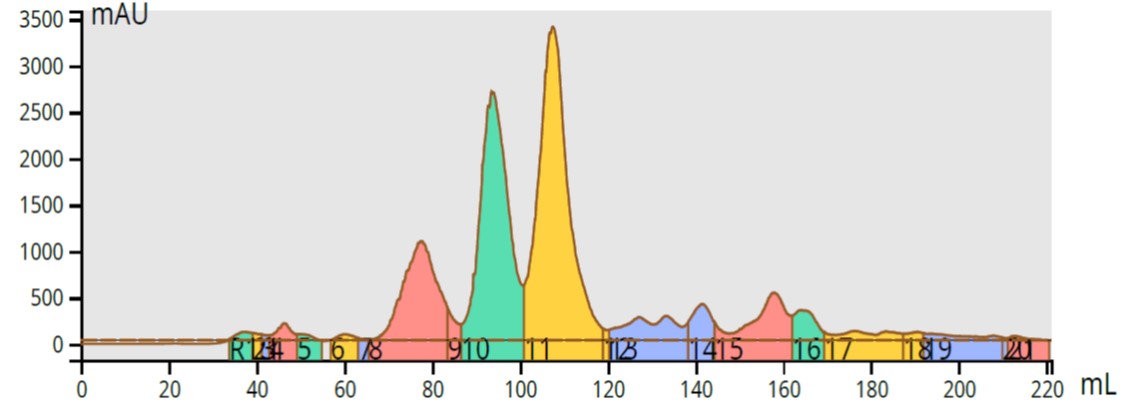
Figure 3. Automated flash purification of a highly complex reaction mixture (Biotage Selekt with a Sfar flash column).
Some automated flash purification systems have multiple detection options including both UV and either an evaporative light-scattering detector (ELSD) or mass detector. These optional detectors can help find hidden impurities or compounds not detectable by UV, Figure 4.
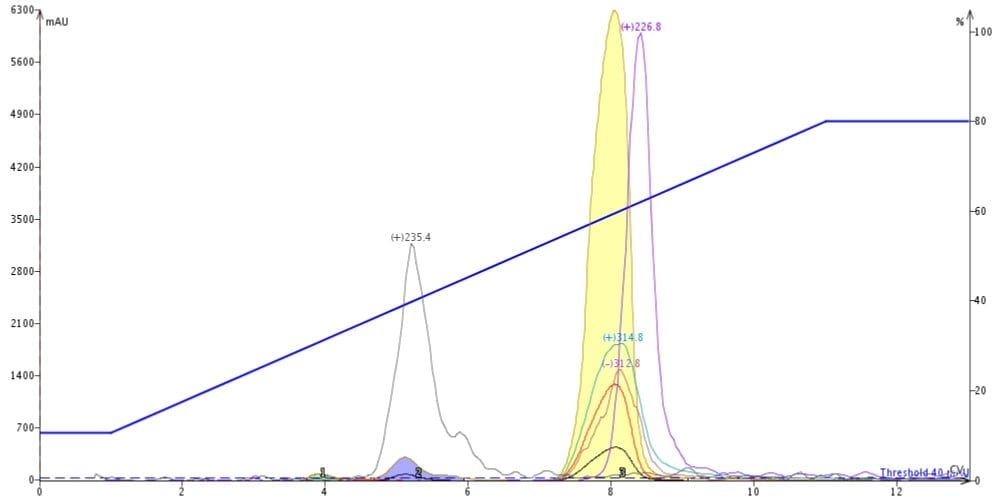
Figure 4. Automated flash chromatography with mass detection (Biotage Dalton 2000) can find hidden impurities.
Once your product is purified, it can then be used as a reagent for the next synthesis step and the fun continues…
Want to learn more about organic synthesis tools? We've got a specific breakdown on getting a flash chromatography system here:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership