Welcome to a second installment of the Biotage Cannabis Application development flash blog. The first post, dated August 20 2016, outlined an orthogonal approach to isolating cannabinoids from winterized extract.
Today we investigate the “hot” topic of pesticide elimination from extract. Specifically – Myclobutanil remediation.
Myclobutanil, aka Myclo and commercially known as Eagle 20®, is a commonly used fungicide applied in agriculture to reduce/prevent powdery mildew and leaf spots, Figure 1.
Without going into detail about the pesticide's toxicity, it’s needless to say that the presence of Myclo in consumer products is highly regulated by state public health agencies. Depending on your geographical location, acceptable limits of this pesticide can range in the low PPM or even PPB concentrations, Table 1.
| State | OR | CA | NV |
| Limits | 0.2 ppm | 0.02 ppm | 9 ppm |
Table 1. State action limits for myclobutanil
What can be done to remove Myclo from cannabis oils? In this experiment, we attempt to remove a significant amount of Myclobutanil from THC distillate by way of flash chromatography. Flash chromatography has already been shown useful to isolate cannabinoids.

Figure 1. Myclobutanil chemical structure
To perform the study we started with about 50 grams of THC distillate and then added a calculated amount of Eagle 20 to formulate a Myclo concentration of a whopping 111.5 ppm. Yep, that’s a lot! When present, typical Myclo concentrations run in the single digit to low 10’s ppm range.
For the flash chromatography portion of the experiment we used a Biotage Isolera™ Spektra One flash system, a 30 gram Biotage® SNAP Ultra C18 column and a 25 gram Biotage SNAP KP-Sil column.
For purification fraction analysis a Shimadzu GCMS-QP2020 was used to determine Myclo content.
First, we ran a normal-phase purification experiment utilizing the 25g SNAP KP-Sil column loaded with 2.5 grams of Myclo-spiked sample. That is a mass load ratio (sample/column) of 10%. An optimized step gradient with common organic solvents provided a single peak comprised mostly of THC cannabinoid, Figure 2. Fractions 2, 3 were pooled and evaporated to remove any trace amounts of organic solvent. The dried sample was solvated in ACN and injected into the GC-MS system.
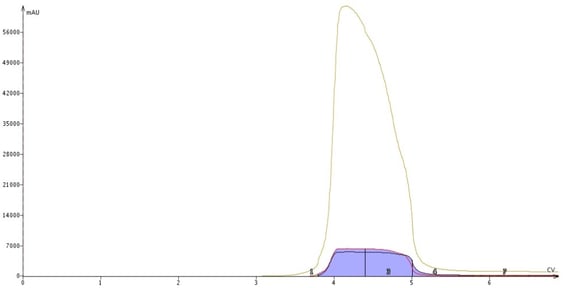
Figure 2. Normal-phase flash chromatography of a THC extract containing 111.5 ppm of Myclobutanil. The peak seen here contains purified cannabinoids and minimal amount of other compounds.
A flash purification investigation wouldn’t be complete without including reversed-phase (RP) chromatography. For this experiment, we utilized a 30g SNAP Ultra C18 column loaded with Myclo-spiked sample to a mass/mass ratio of 3%.
Sometimes chromatographic separation is enhanced with a pH modifier. To explore this potential, we decided to split the RP purification experiments into two runs and evaluate both neutral and acidic gradients. For the acidic gradient, formic acid was added to the solvent at a concentration of 1%. Optimized linear gradients of ethanol+water and ethanol+water+formic acid provided single peaks of mostly THC cannabinoid, Figure 3. Fractions 6-11 were pooled in both runs and evaporated to dryness. The resulting dried sample was then solvated in ACN and injected into the GC-MS system.
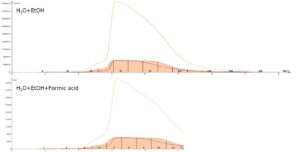
Figure 3. Reversed-phase purification of Myclobutanil-spiked extract. Top - water/ethanol gradient, bottom - water/ethanol/formic acid gradient. The chromatograms indicate no visible impact from the formic acid.
Fractions from all purification runs were analyzed for content and purity by GC-MS, Table 2. This data shows that flash chromatography using neutral pH reversed-phase flash chromatography removed 99.7% of the myclobutanil from the sample and normal-phase flash removed 99.6%. The data show adding formic acid did not improve myclobutanil remediation efficiency.
| Step | [Myclobutanil] ppm |
| Spiked Sample | 111.54 |
| Clearance post-chromatography | |
| Normal-phase | 0.44 |
| Reversed-phase | 0.31 |
| Reversed-phase, acidic | 1.09 |
Table 2. Myclobutanil remediation efficiency results
The 99 plus percent clearance in both applications is quite an accomplishment provided the very high starting Myclo concentration in our distillate sample. It’s common chromatographic knowledge that sample load ratios can impact separation efficiency. What if our sample had only 10 ppm concentration of Myclo instead of 111 ppm? What if we utilized the orthogonal approach and ran both normal- and reversed-phase separations in step sequence to remove Myclo from a single sample? Can scavengers resins be utilized to reduce/remove pesticides? I encourage you to stay tuned to our Cannabis Blog as we’ve got much more planned in the very near future.
Biotage would like to thank NorCal Analytical for their contributions in these experiments.
So, how have you addressed removing pesticides for plant extracts? Please share so that our readers can learn or check out our infographic on cannabis below!

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership