Flash chromatography is a chemical separation technique used to purify chemical mixtures. Because it is a purification technology, the process is also referred to as flash purification.
Whether you are synthesizing novel molecules or trying to isolate a natural product, flash chromatography is a “must do” part of your workflow. Synthetic reactions almost always have reaction by-products and unreacted starting materials that need removal prior to the next step in the reaction. If the target compound is not purified, then the risk of creating even more undesirable by-products in subsequent syntheses increases.
The same is true for natural product research. When you extract your biomass or fermentation broth, not only does your target get extracted but so do other compounds with similar solubility. Co-extractant impurity removal/isolation is critical to enable individual compound testing that will generate meaningful results.
The principal behind this separation technology is simple – compounds in solution will, based on their particular chemistry, separate from each other due to their polarity differences, given the right conditions and technique. These chemical differences are based on each compound’s solubility in a particular solvent. Compounds that have lower solubility can be removed from those with greater solubility using the same solvent.
The separation process uses a column filled with a pure, solid medium such as synthetic silica onto which the chemical mixture is introduced. After the mixture is added to the top of the column, solvent is introduced into the column using a pump. Those compounds with higher solubility will migrate through the column faster than those with less solubility and can be collected individually. The collected materials will be purer than when they were in the original crude mixture.
Figure 1 shows how flash chromatography separated the various compounds in a natural product extract. The target compound is the large blue peak, which is separated from other compounds in the extract and collected into two fractions (#6 and 7).
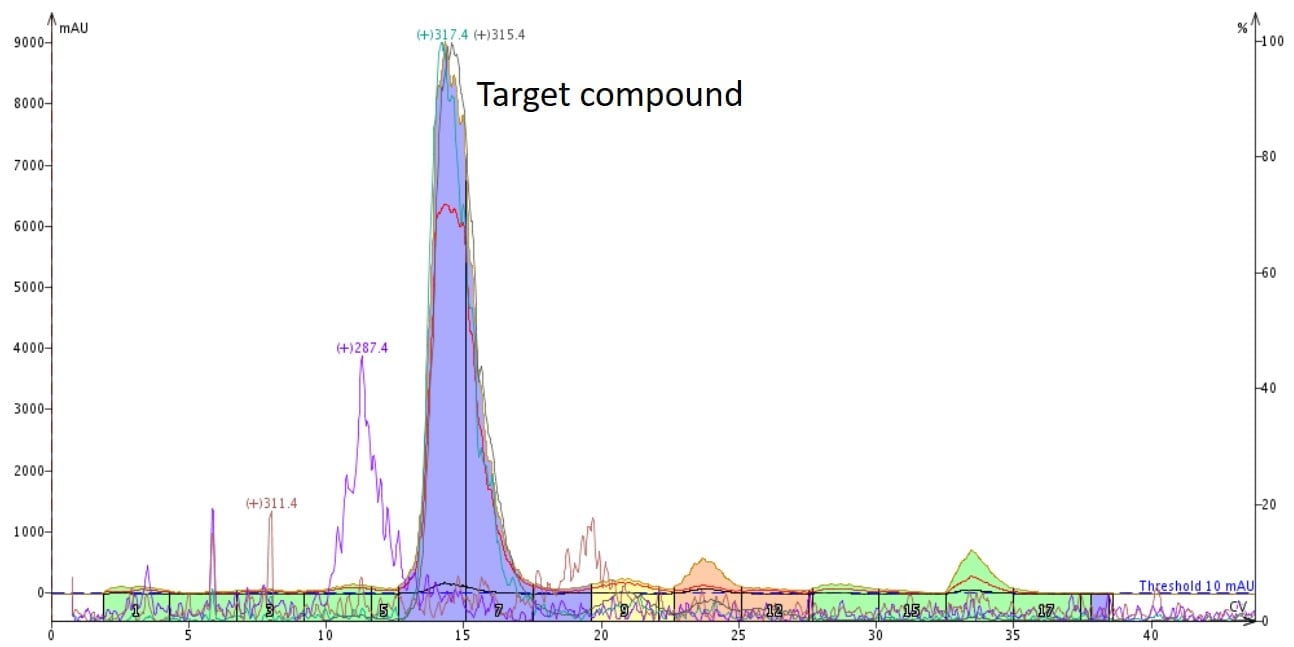
Figure 1. Natural product extract flash chromatography separated several compounds including the targeted molecule (large blue peak). The target compound was collected in fractions 6 and 7.
Figure 2 shows the purification results for the fractions 6 and 7 (target compound). Virtually all of the other compounds in the extract are removed from the desired compound indicating it is pure.
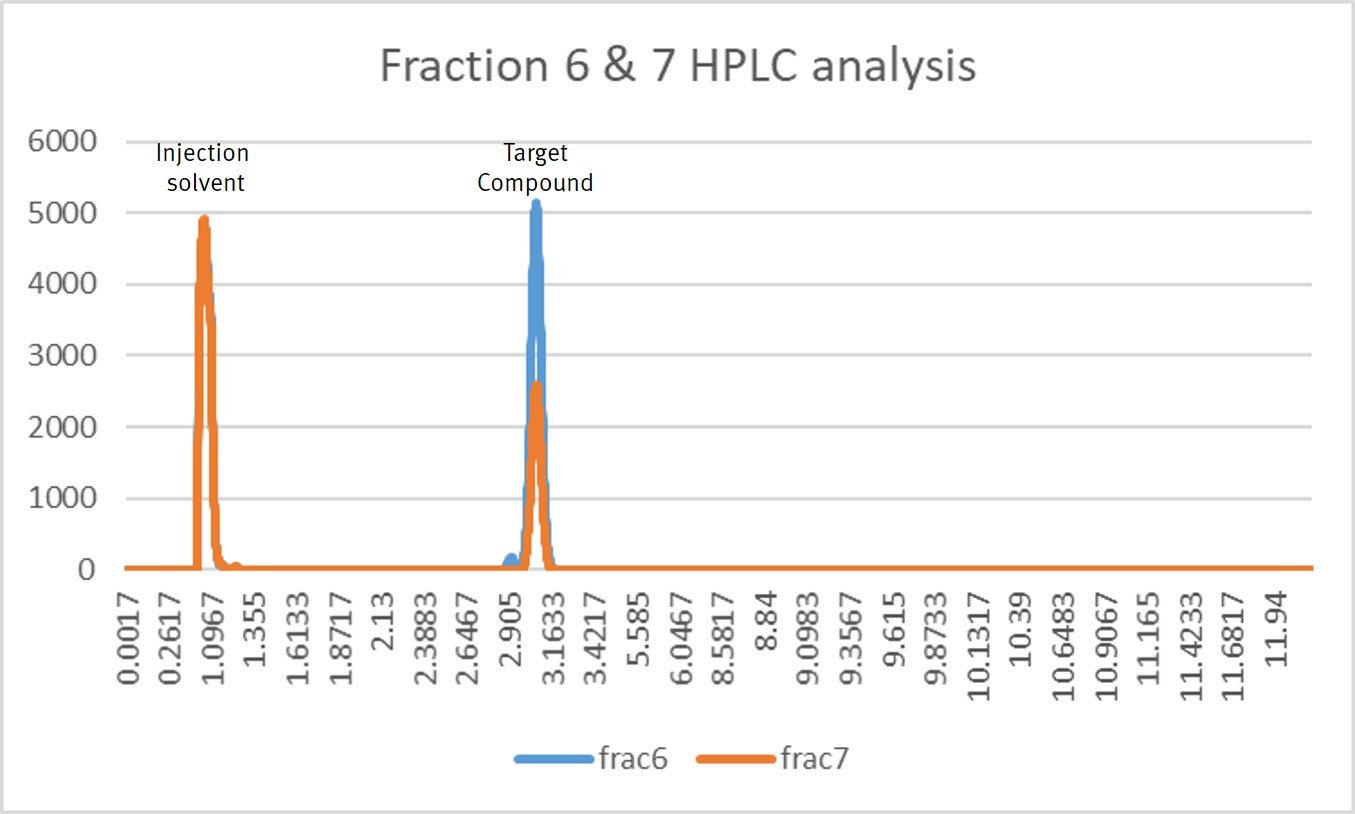
Figure 2. Target compound flash chromatography fraction 6 and 7 analysis by HPLC shows the desired compound is nearly 100% pure.
In order to isolate your desired compound or compounds from crude mixtures, use flash chromatography to help you achieve that goal.
To learn more about flash chromatography, please read our latest white paper!

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership