UV detection and fractionation is ubiquitous in flash chromatography. It is the default methodology used to detect and collect eluting compounds. Today’s flash chromatography systems offer UV-triggered fractionation on one, two, or a range of wavelengths in order to either increase fractionation specificity, yield, or increase sensitivity.
In this post, I will discuss the value of incorporating fractionation based on a wavelength range and two selectable individual wavelengths.
Flash chromatography has been used for natural product and reaction mixture purification for over a century. Most early compound detection and fractionation was based on color (hence the word chrom in chromatography). However, for non-colored materials chemists would base their fraction collection by time or volume and then analyze the collected fractions by TLC with either UV or staining/post-separation reaction for compound detection.
As flash chromatography has matured, flash system manufactures added UV detection into available chromatography systems to help chemists identify collection vessels containing UV absorbent compounds. These systems later were able to control automated fraction collectors such that they used the detector’s response to trigger fractionation based on a single wavelength (usually 254 nm). Later, diode array detectors became standard in automated flash systems enabling tunable fraction collection on one or two different wavelengths – a technique commonly used today.
Full wavelength range detection is now standard with most flash systems. Chemists can tune the wavelength range to maximize sensitivity (very helpful for low wavelength absorbing compounds). With this capability, some systems also offer the option to select individual wavelengths for monitoring or fractionation – it’s nice to have this flexibility.
While detection/fractionation using one or two individual wavelengths isolates individual eluting compounds absorbing at those specified wavelengths, there will be some sample loss. This, of course, is due to the fact that compounds absorb UV and/or visible light over a wavelength range, not any one or two specific wavelengths, Figure 1.
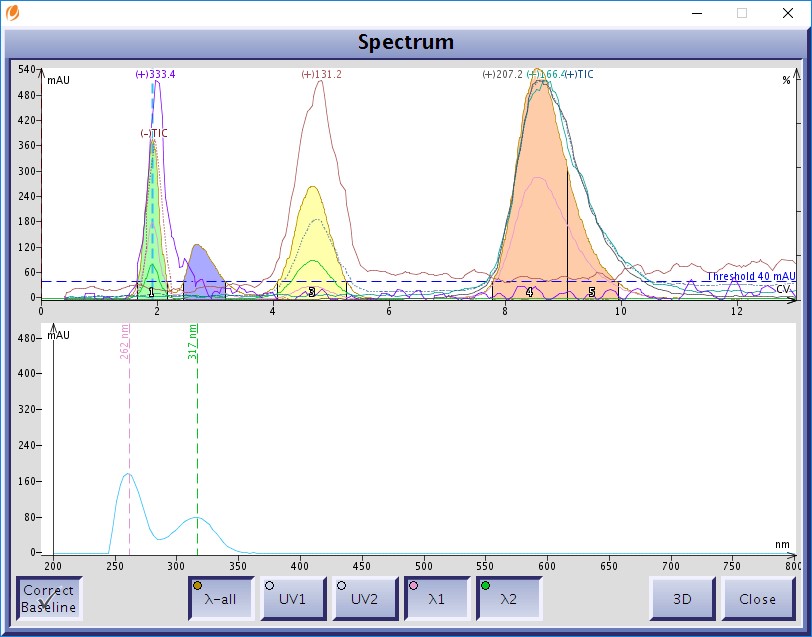
Figure 1. Diode array UV-vis spectra of a compound purified by flash chromatography. this compound has two UV maxima at 262 nm and 317 nm. Most organic molecules and more than on UV maxima.
Most chemists I have talked with about flash chromatography tell me compound yield is more important than compound purity (these are typically intermediates being synthesized), but that they typically need 80%+ purity. Depending on how well a compound absorbs light at a specific wavelength, obtaining a high purification yield and purity for their compound may be a challenge, Figure 2. In cases like this where one compound co-elutes with another that has a different UV spectrum, only part of the first eluting peak is isolated from other compounds (~ 6 ml, green), while the second part (yellow) is mixed.
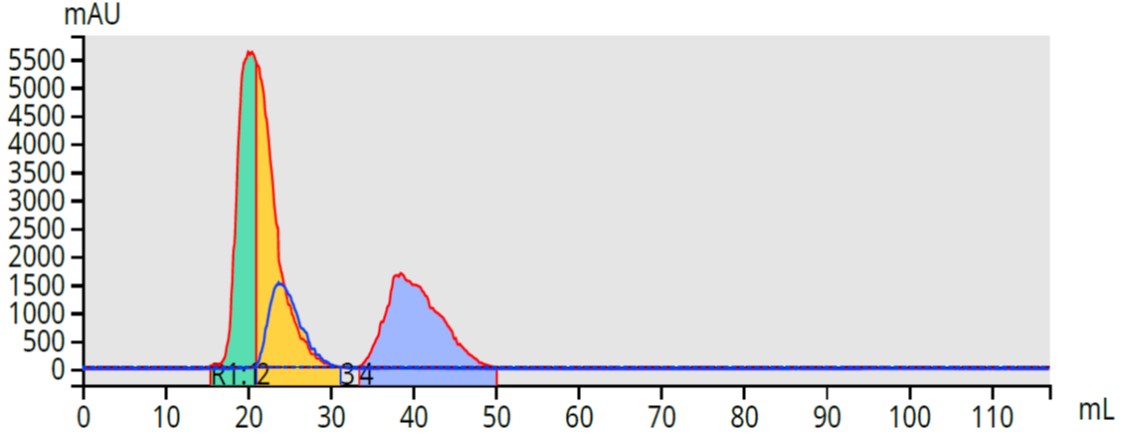
Figure 2. Three-component purification with dual-wavelength fractionation. Two compounds co-elute but are partially purified using fractionation with two different wavelengths. Only the first 5-ml of the first peak is pure.
All-wavelength detection and fractionation was developed to solve this issue. With the Biotage® Selekt system, any wavelength range between 200-400 nm (variable UV) or 198-810 nm (UV-vis) can be set. The broader wavelength ranges provide increased sensitivity as the detector response is based on the total absorbance over the range rather than the absorbance at a single wavelength. This is important because increased sensitivity = increased yield.
While this increased sensitivity improves yield, it can have a negative impact on purity. This is possible if there are two or more compounds eluting very closely that have different UV specta. Because the separation is poor and the detection wavelength range is broad, both compounds are encompassed in a single peak, Figure 3. The result is a fraction with high yield and no purity.
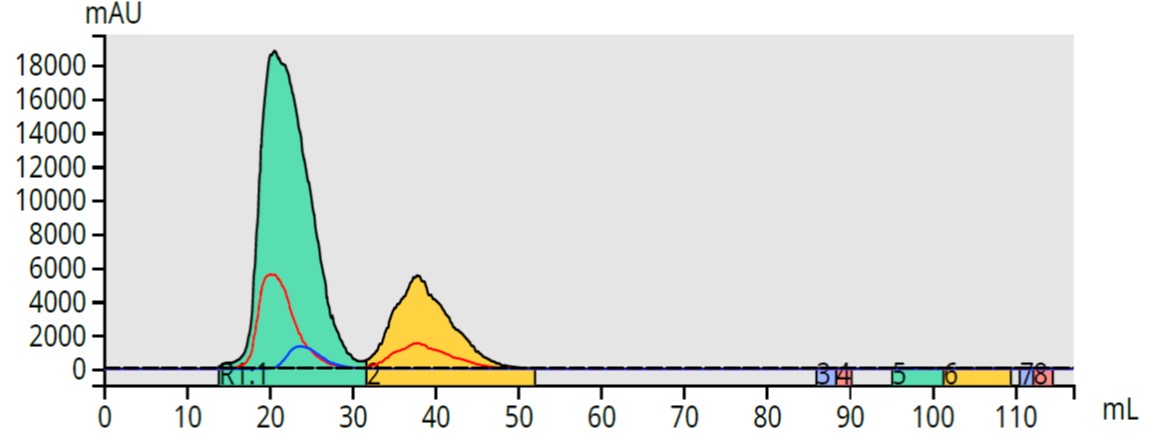
Figure 3. All-wavelength detection (200-400 nm) of a sample containing three compounds fractionates only two peaks. Two of the compounds partially co-elute and their combined absorbance does not provide individual compound fractionation.
If the purification conditions cannot be adjusted to perform a better separation, then using a combination of collection on all wavelengths plus triggering fractionation based on two individual wavelengths, provides improvements in fractionation specificity which increases both purity as well as yield, Figure 4.
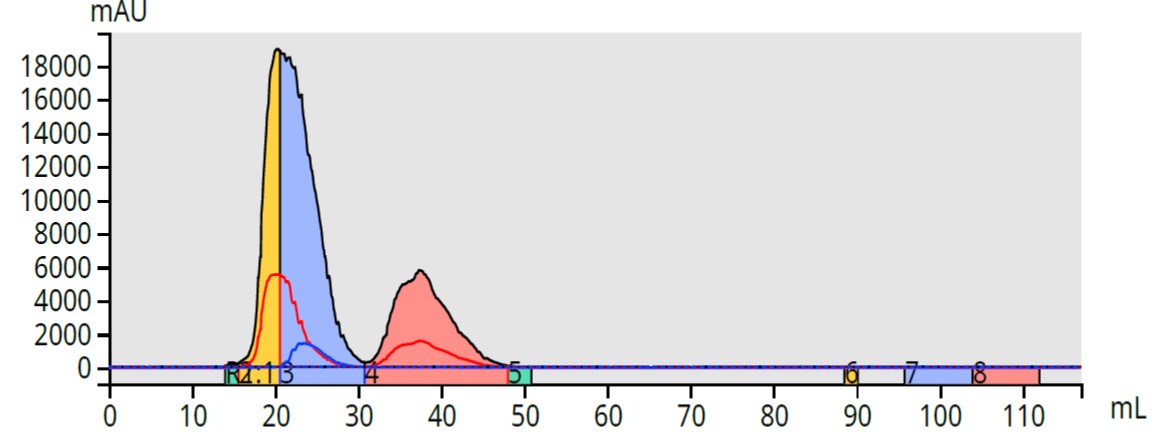
Figure 4. Fractionation using a combination of two selected wavelengths plus all-wavelength diode array (200-400 nm) provides specificity and increases both yield and purity. The first eluting peak volume (yellow +green) increased from 5-mL (Figure 1) to 8-mL, a yield improvement of 60%.
So, when using automated flash chromatography with diode array detector, fractionate using multiple UV responses, individual wavelengths + all-wavelength summation, to improve both purification yield and purity.
To learn more about flash chromatography, download my white paper here:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership