Method transfer from reversed-phase TLC (thin layer chromatography) to reversed-phase flash column chromatography can be very challenging. Because of this, I often recommend using HPLC for reversed-phase flash chromatography method development. This really is a straight-forward process if you start with the right HPLC column and know a little about your HPLC system's detector.
In this post, I will share some tips on how to develop a reversed-phase flash purification method using HPLC.
In a previous post I discussed the challenges that reversed phase TLC poses for flash column purification, the primary issue being that surface wetting of the C18 stationary phase provides inconsistent prediction of chromatography conditions.
So, with reversed-phase TLC not being a reliable method development tool for flash column chromatography what other options are available? Well, I normally recommend reversed-phase HPLC with a similar bonded phase chemistry, e.g. C18.
Method development using HPLC
Now that you know the reasoning behind why method development on HPLC for reversed-phase flash chromatography is preferred, I need to explain how to do the conversion.
Simply put, the process is to create a gradient on an HPLC system and then to duplicate it on a flash system. It is best to use a HPLC column that is packed with the same media as the flash column (Biotage offers HPLC columns packed with their flash C18 media, they are known as scaling columns). Using a scaling column for method development eliminates any separation quality differences related to particle size and the resulting resolution/efficiency impact on the separation.
Using an off-the-shelf, commercially available HPLC column will certainly point you in the right direction regarding the gradient method but due to media particle size and potential selectivity differences, the results may not translate as well to flash as you might want.
To illustrate the challenges associated with using a generic reversed-phase HPLC column for method development, I conducted an experiment where I used a commercially available HPLC column to develop a suitable separation method and then transferred the exact method to flash chromatography.
Table 1 shows the HPLC gradient, which was based on time, in tabular form.
| Time | %MeOH | Flow rate (mL/min) |
| 0 | 55 | 1 |
| 4 | 55 | 1 |
| 24 | 90 | 1 |
| 26 | 90 | 1 |
| 26 | 100 | 1 |
| 32 | 100 | 1 |
Table 1. Final HPLC gradient
In Figure 1, both the gradient and results are shown in graphical form. For the HPLC work a Phenomenex Gemini® (4.6 x 150 mm, 5 µm) was used and found to provide adequate separation results for transferring the method to reversed-phase flash chromatography
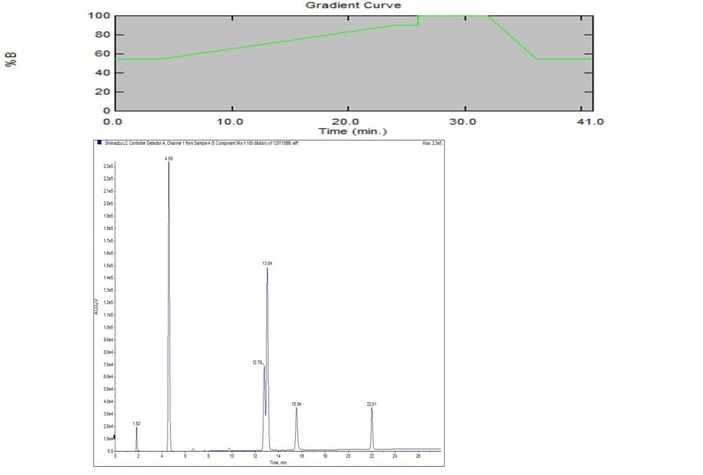
To accomplish this conversion I made note of the HPLC retention time for peak 1 (1.82 min) which indicates the void time. Since the flow rate was 1 mL/min that means the void volume for the column is 1.82 mL which is equal to 1 column volume (CV). Converting the HPLC method from time to column volumes eliminates the need to know column size and flow rate when duplicating it on a flash system; Table 2 displays the gradient in Table 1 in terms of column volumes.
| CV | %MeOH | Flow rate (mL/min) |
| 0 | 55 | 1 |
| 2 | 55 | 1 |
| 12 | 90 | 1 |
| 12 | 90 | 1 |
| 13.01 | 100 | 1 |
| 16 | 100 | 1 |
Table 2. HPLC method converted from time to column volumes
I programmed the flash system (I used an Isolera One™ system) with the CV-based gradient and selected a 12-gram SNAP Ultra C18™ (Biotage) to perform the purification, Figure 2.
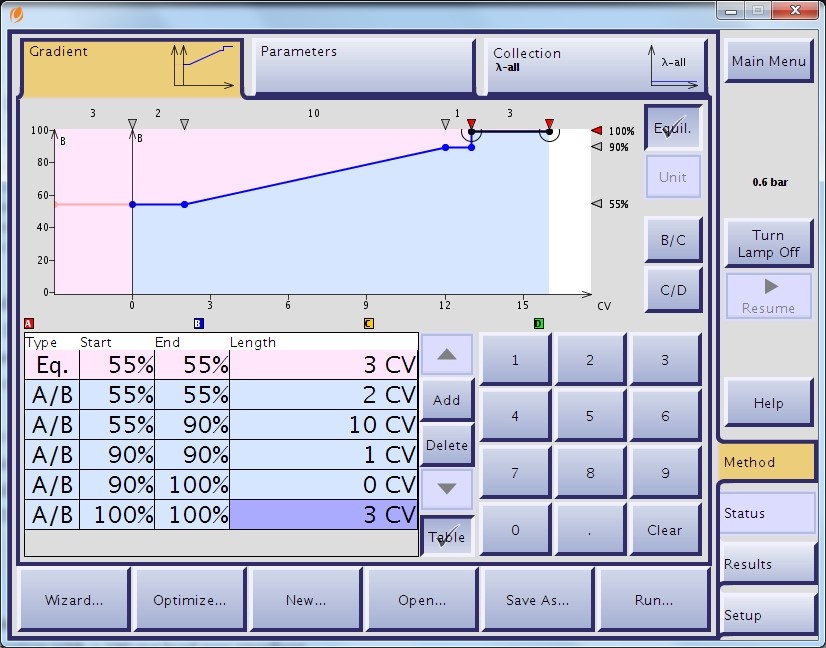 Figure 2. Reversed phase flash method based on HPLC gradient.
Figure 2. Reversed phase flash method based on HPLC gradient.
For the flash purification I loaded 100 mg of the sample. The results show a full separation between each compound but with some slightly different selectivity compared to the HPLC column. In fact, the flash column actually separates the third eluting peak (yellow) better than the HPLC column while the separation between peaks 4 and 5 is less than the HPLC, Figure 3.
So, what do these results really tell us? Well, the results indicate that the generic reversed-phase HPLC column was able to approximate the potential flash cartridge performance but it was not a direct correlation. This is why the use of a scaling column packed with the same media as the flash cartridge makes sense - the selectivity and resolving power will be identical making scale-up to flash much more straight-forward.
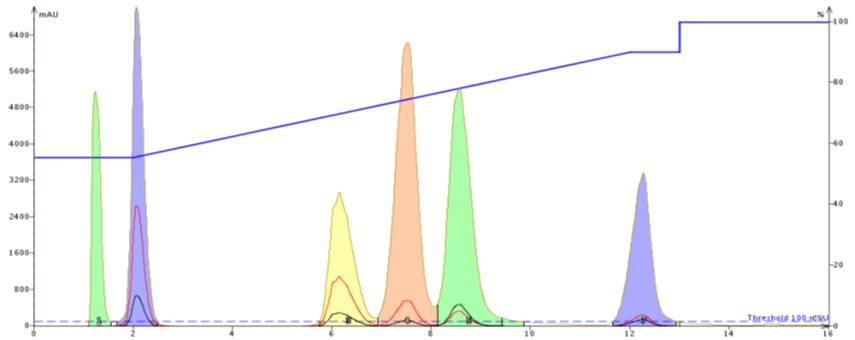 Figure 3. Flash reversed-phase purification using the HPLC-developed gradient. The gradient segments are matched in terms of column volumes and the results are similar but not identical to the HPLC column. The flash column is a 12-gram SNAP Ultra C18 operated at 20 mL/min with a sample load of 100 mg.
Figure 3. Flash reversed-phase purification using the HPLC-developed gradient. The gradient segments are matched in terms of column volumes and the results are similar but not identical to the HPLC column. The flash column is a 12-gram SNAP Ultra C18 operated at 20 mL/min with a sample load of 100 mg.
Scaling column based method development
To start, set some basic parameters then modify the gradient start and end points as needed to obtain a suitable separation. In Table 3, below, I show some example starting points for two Biotage C18 media columns.
| KP-C18-HS scaling column | Ultra C18 scaling column | |
| Column size (mm x mm) | 4.6 x 250 | 4.6 x 250 |
| Particle size (um) | 50 | 25 |
| Flow rate (mL/min) | 1 | 1 |
| Linear velocity (cm/min) | 7.5 | 6.7 |
| Detection | Diode array | Diode array |
| Run time, gradient (min) | 43 | 49 |
| Solvent A | Water | Water |
| Solvent B | Methanol or acetonitrile | Methanol or acetonitrile |
| Equilibration | 10% B for 10 min. (3CV) | 10% B for 11.2 min. (3CV) |
| Gradient segment 1 | 10% B for 3.3 min. (1 CV) | 10% B for 3.7 min. (1 CV) |
| Gradient segment 2 | 10% to 100% B in 33.2 min (10 CV) | 10% to 100% B in 37.4 min (10 CV) |
| Gradient segment 3 | 100% B for 6.6 min. (2 CV) | 100% B for 7.5 min. (2 CV) |
| Sample size (mg) | less than 1 | less than 1 |
Table 3. Suggested reversed-phase method development starting points
Basically, create a 3-segment linear gradient based on column volumes using the scaling column. The scaling column's column volume is the void time (t0) multiplied by the flow rate.
Equilibration 10% MeOH (or MeCN) for 3 CV
Segment 1 10% MeOH (or MeCN) for 1 CV
Segment 2 10% MeOH (or MeCN) to 100% in 10 CV
Segment 3 100% MeOH (or MeCN) for 2 CV
If your compounds elute too early, reduce the end % B to 50% and repeat (do not change the run time or flow rates).
If your compounds elute late in the gradient, increase the start % B to 50% and repeat (do not change the run time or flow rates).
Continue this process until you have an acceptable separation then follow steps 1 through 6.
Challenge the column’s loading capacity until your target compound has a resolution (Rs) ~1 from its nearest neighbors, this will be your loading limit which can be scaled up to any size flash cartridge. Most HPLC systems have software that will calculate resolution.
- Find out what the HPLC system’s flow cell path length is and divide it by the path length of your flash system’s detector (0.3 mm on all Isolera systems). This will help compensate your flash loading for the more sensitive analytical HPLC detection.
- Multiply your scaling column load by the number calculated above to give you an estimated flash system load for the scaling column.
- To choose the proper cartridge size for the amount of material you need to purify, check Table 4.
- On your flash system create the same method in CV choosing the appropriate cartridge media and size for your purification.
- Set your flash flow rate to match the scaling column linear velocity using Table 5 below.
Table 4. Scale up factors for reversed-phase
| Column size | Scale factor |
| Scaling column | 1 |
| 12 gram | 4.6 |
| 30 gram | 11.5 |
| 60 gram | 23 |
| 120 gram | 46 |
| 400 gram | 153 |
Table 5. Flash column flow rates with equivalent linear velocities
| Column size | KP-C18-HS flow rate (mL/min) | Ultra C18 flow rate (mL/min) |
| Scaling column | 0.5 | 0.5 |
| 12 gram | 10 | 12 |
| 30 gram | 17 | 24 |
| 60 gram | 30 | 40 |
| 120 gram | 30 | 40 |
| 400 gram | 105 | 135 |
If you follow this process you should find success moving a reversed-phase HPLC method to flash.
Share your reversed-phase flash method development techniques with the rest of the readers and suggest other topics you would like to see discussed.
Interested in learning more about flash chromatography? Please click the link below to my webinar Inspiring Productivity with Modern Flash Chromatography

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership