Evaporative Light-Scattering Detection, or ELSD for short, is a technology used with liquid chromatography to see UV-transparent (and UV-absorbing) compounds. In a previous post I talked about some applications where ELSD is not only useful, but required.
In this post, I will explain how an ELSD is configured and functions.
An ELS detector is quite different than most other detectors used in chromatographic applications. It does not require that compounds have any specific functionality or ionization capabilities. Its only requirements are that the compound does not evaporate under the conditions set on the detector and the solvent does. These conditions include setting evaporation temperature, inert gas flow, sample inlet flow, and sensitivity level.
So, what is the basis for the ELSD’s operation? Well, it is rather simple actually and can be explained in the following steps.
- -The chromatographic cartridge effluent is split so that a small portion is directed to the ELSD and the majority carries on to the fraction collector.
- -The ELSD effluent is mixed with inert gas (usually nitrogen) and heated to a specified temperature; this helps to evaporate the effluent.
- -The evaporated solvent flow then moves into a heated chamber, sometimes referred to as a drift tube, where the vapor is heated further (sometimes at a higher temperature) to ensure the solvent evaporates. It is during this evaporation process that the compounds form “air-born particles”.
- -The “dried” particles are then detected by either a laser or LED light source which triggers fraction collection.
-The particles produced at the end of the drift tube are what are to be detected. so virtually any substance that will not evaporate in the drift tube and forms a particle or even an oil will be detected.
To help visualize this process I have included a graphic, which is representative of the Biotage® A-120 ELSD, Figure 1.
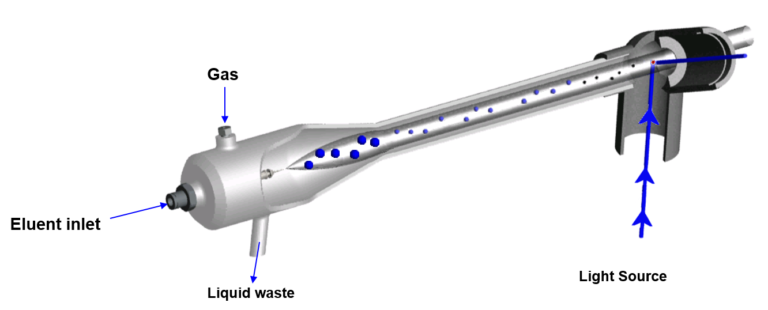
Figure 1. Inner workings of an ELSD. Cartridge effluent is split enabling a small portion to enter the ELSD where an aerosol is generated and the solvent evaporated leaving behind particles detectable by a light source.
If you are interested in learning more about flash chromatography, please download our white paper Successful Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership