Using a “dry” loading technique with flash chromatography typically improves compound purity and overall separation quality compared to liquid loading. The reasons for this I have prophesized previously and include:
- -Pre-purification sample concentration which reduces sample dilution and bandspreading
- -Removing the injection solvent effect in which the injection solvent’s volume displaces an equal volume in the column thus reducing the amount of sorbent available to perform the separation
- The injection solvent’s polarity reduces the amount of solute/sorbent attraction leading to early elution, misshaped peaks and poor resolution
If you are not familiar with dry loading, it is a sample loading technique that uses a blend of reaction mix/extract and sorbent. The sample solution is mixed with sorbent, dried, and then packed into a separate vessel that is then attached to the purification column, Figure 1. Once the purification method starts, the mobile phase solvent dissolves compounds based on their solubility and carries them into the purification column where the separation takes place.
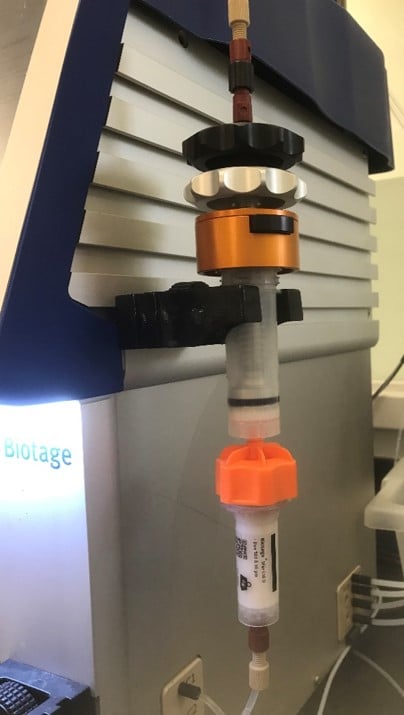
Figure 1. Example of an external dry load vessel (top) attached to a purification column (bottom).
Historically, most flash chromatography has been normal-phase (polar silica column, low to mid polarity organic solvents). Those using this flash purification mode usually use silica or diatomaceous earth (e.g. Celite®) as dry load sorbents.
The past 10+ years has seen great growth in reversed-phase flash chromatography. While reversed-phase uses different column media (non-polar surface) with polar, water miscible solvents, it too has the same sample load issues mentioned above. With that being the case, which dry load sorbents are best for reversed-phase flash chromatography?
Curious about this I tested three Biotage brand dry loading sorbents (KP-Sphere™ silica, ISOLUTE® HM-N (a refined diatomaceous earth), and KP-C18-HS™) comparing them to liquid loading.
While the separation was quite easy (butyl and methyl paraben), the injection technique and sorbent selection differences should still be discernable.
To start, I used a 6 gram Biotage® Sfär C18 Duo column with a 25% to 80% acetonitrile gradient. A 0.5 mL injection size (acetone sample solvent) provided a 100 mg load. The two parabens separated but with broad bands for methyl (27 mL) and butyl (23 mL) paraben, Figure 2.
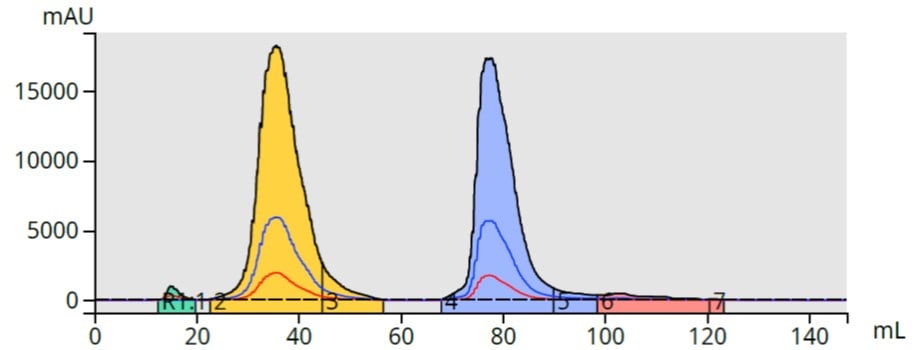
Figure 2. Reversed-phase paraben separation using a liquid load (0.5 mL).
The same sample, load, and gradient method were then used to see how well the dry load sorbents, in the Biotage Samplet format, performed vs. liquid loading and each other, Figure 3.

Figure 3. A Samplet cartridge ready for installation inside a Biotage Sfar Duo column.
Using the same sorbent for dry loading as for the purification is logical (matching media eliminates a variable). I found this approach worked and greatly reducing methyl paraben’s elution volume from 27 mL to 17 mL (37%), Figure 4. Later eluting butyl paraben’s elution volume remained about the same.
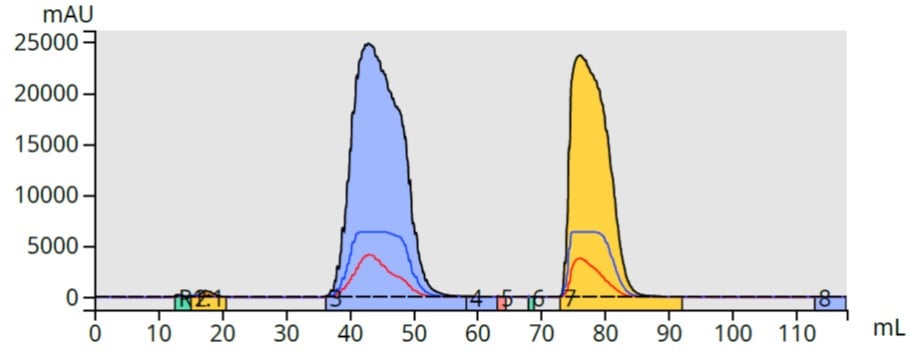
Figure 4. Paraben purification using a C18 filled Samplet cartridge greatly reduces fraction volume for methyl paraben (blue) compared to the liquid injection.
The purification using HM-N media provided a separation similar to the liquid load with fully separated peaks collected in similarly large volume fractions, Figure 5. The higher volume fractions are likely due to the larger HM-N media particle size.
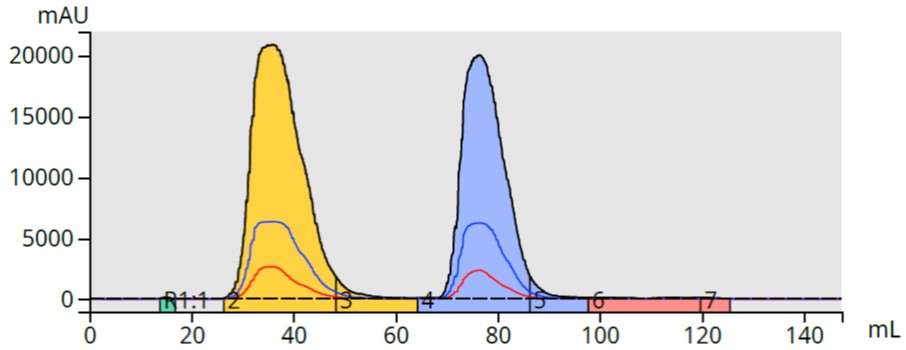
Figure 5. Using the refined diatomaceous earth dry load media provided similar collection volumes for the two parabens.
Lastly, I used silica as the dry load sorbent. Though counter intuitive because of the extreme polarity difference with the column media, the results show an excellent separation with tight elution bands. While the methyl paraben volumes were about the same, butyl paraben was collected in about 4 mL less solvent than with the C18 dry load, Figure 6.
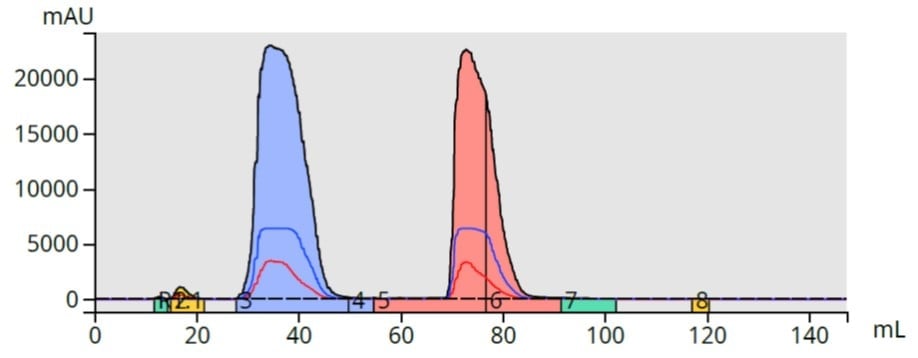
Figure 6. Purification using polar silica as the dry load sorbent allowed butyl paraben (pink) to elute in minimal volume (~18 mL).
So, why does silica work well as a dry load medium for reversed-phase? My theory is butyl paraben's greater hydrophobicity (compared to methyl paraben) decreased its affinity for the silica dry load sorbent and allowed it to elute quicker and in a higher concentration than methyl paraben.
The bottom line, however, is that you can use most any sorbent for reversed-phase dry loading but, with compounds similar to those used in this study, the best results will be obtained using either C18 or silica.
If you want to learn more about the options with Sfär, check out the page below:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership