This is an interesting question that does not have a straightforward answer. In fact, there are many materials that are potentially useful sorbents for dry loading crude mixtures. Some of the more popular are silica, diatomaceous earth (e.g. ISOLUTE® HM-N, Celite®), alumina, and Florisil®. The sorbent choice can influence your purification results because each of the available media have different chemistry and capacity.In most cases, sample/sorbent reactivity really is not a major concern, though it can occur. What is important is the sorbent’s capacity to adsorb/absorb all of your sample and the ratio of your crude sample to the amount of dry load sorbent.
In this post, I discuss the ability of three different sorbents (silica (Biotage® KP-Sphere), ISOLUTE HM-N (a diatomaceous earth), and Florisil® (a magnesium silicate) to adsorb/absorb a reaction mixture.
As you know, organic synthesis can be either very straightforward yielding your product with minimal by-products or complex with many “impurities” requiring removal, typically by flash chromatography.
In the case of the latter, the purification approach should be one well thought out. A simple liquid injection may be convenient but not yield the desired result. To illustrate this, I performed a reaction in acetone that yielded several by-products.
To purify the reaction mixture, I used a flash method starting at 20% ethyl acetate and ending at 40% with a 5-gram high-performance Biotage® Sfär column and a Biotage® Selekt flash system with a load of 0.4 mL (100 mg). The purification showed promise but results were less than acceptable with minimal compound resolution, Figure 1.
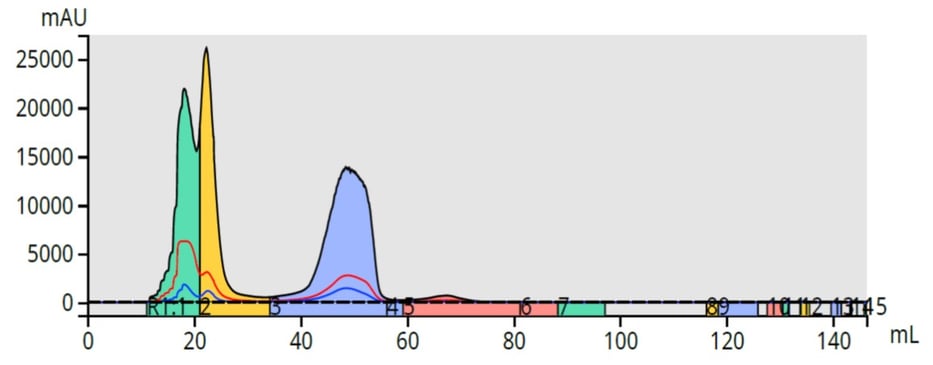
Figure 1. Flash purification results using liquid loading (0.4 mL) and a 5 gram flash column shows an incomplete separation with four detected compounds.
The poor results were due to liquid loading the acetone-solvated reaction mix. It is important to understand that even small injection volumes can cause chromatographic issues.
When your chemistry is complex or mixture dissolved in a strong solvent, you should consider dry loading. However, this gets back to the original question – which media should be used?
Well, I thought I would evaluate three media I had available for dry loading – silica, HM-N, and Florisil. For this study, I used a sample/sorbent ratio of one part crude (100 mg) to four parts sorbent (400 mg) for my purification. This ratio is one I have found to work well. The results show that for this particular reaction mixture silica provided the best results, Figure 2.
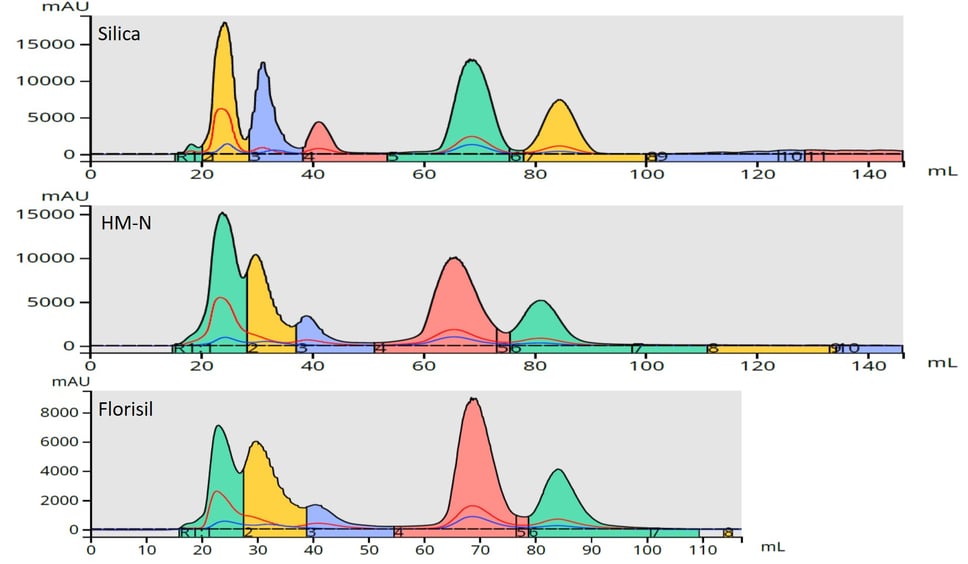
Figure 2. Dry load flash chromatography results with different dry load sorbents show various improvement levels. Top - silica provides the best results likely due to its very high surface area and adsorptivity. Middle - diatomaceous earth. Bottom - Florisil.
The reason that silica performs the best is likely related to its high surface area (~750 m2/g), which gives it increased capacity. Silica, however, may not be the right media for your reaction mix purification so you may want to perform your own study.
The point of this study is to suggest that spending a little time to evaluate dry loading sorbents for your crude material purification can provide you with the best possible results.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership