Mass-directed purification, whether with a preparative HPLC or a bench-top flash system, is quickly gaining interest in the peptide purification space. The simple fact is that using a specific mass, rather that UV absorbance, to trigger fraction collection allows for greater confidence in the identity of the collected fraction. Importantly though, this technique can also reduce your time required for purification, by significantly reducing or even eliminating the need for secondary mass analysis of each collected fraction.
When using a bench-top mass-directed flash purification system, a secondary mobile phase or carrier solvent is required to deliver and dilute a fraction of the purification into the mass spectrometer. In today’s post, I’ll discuss how different makeup solvent compositions can affect ionization and ultimately, purification efficiency.
When performing mass analysis on a peptide of interest, there a two ionization strategies that are commonly employed – either electrospray ionization (ESI) or Matrix-Assisted Laser Desorption Ionization (MALDI). I’ll leave a discussion about MALDI mass spectrometry analysis for a later time.
For peptides or other compounds to be detected using ESI methodologies, a charged ion must be formed before entering any part of the mass spectrometer. This typically requires the use of a modifier in the mobile phase which interacts with the compound during the chromatography step or as part of process to introduce the sample into the mass spectrometer (direct injection, for example). This charged species then enters the spectrometer contained in a droplet. The carrier solvent evaporates, which produces a charged particle of formerly dissolved material, allowing detection of the charged species.
The carrier solvent can really be any identity – more often than not acetonitrile or methanol are used. My colleagues, purifying small molecules find that using neat solvents for the carrier solvent and mobile phase solvents enables retention of the ionized states and detection in the mass spectrometer. Attempting to purify peptides without a mobile phase modifier is generally a bad idea from a chromatographic standpoint, but I started to wonder what impact the composition of the carrier solvent has on the efficiency of the ionization. In the case of mass-directed flash chromatography, the sample must be diluted before entering the mass spectrometer so as not to overload the signal. In the Isolera™ Dalton 2000, a 1:1000 split is taken from the column effluent and directed in to the Dalton 2000 mass spectrometer using a carrier solvent.
So, I decided to purify full length GLP-1 using UV detection while simultaneously evaluating the ionization efficiency with changing carrier solvent compositions. Ultimately, it is ionization efficiency that enables mass-directed purification. Full length GLP-1 is a 37-amino acid peptide with an isoelectric point at pH = 5.35 due to 7 acidic residues and 6 basic residues. This peptide has potential for detection as either a positively charged ion at low pH or a negatively charged ion at high pH, Figure 1. The question is, can we see both sets of ions and use those ions to direct the purification of this peptide? Given the extreme dilution of the mobile phase into the carrier solvent, I hypothesized that even a mixed – acidic mobile phase and basic carrier solvent – could potentially provide sufficient ionization for purification. 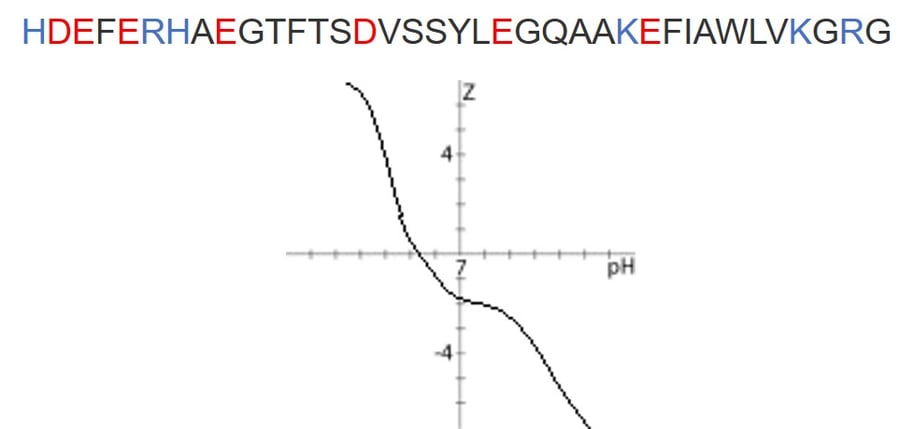
Figure 1. Sequence of the GLP-1 peptide under investigation (top). Acidic and basic residues are highlighted in red and blue respectively. Charge vs. pH plot for GLP-1 sequence above (bottom). At pH 2, the peptide will carry a +6 charge, whereas at pH 10, the peptide will carry a -4 charge.
More often than not, we use acidified mobile phase solvents to purify peptides, but the carrier solvent (95% MeCN:5% water + 0.1% modifier) can have essentially any identity. First, I wanted to test whether or not I could detect the desired peptide ions in either acidic, unmodified, or basic carrier solvent, Figure 2.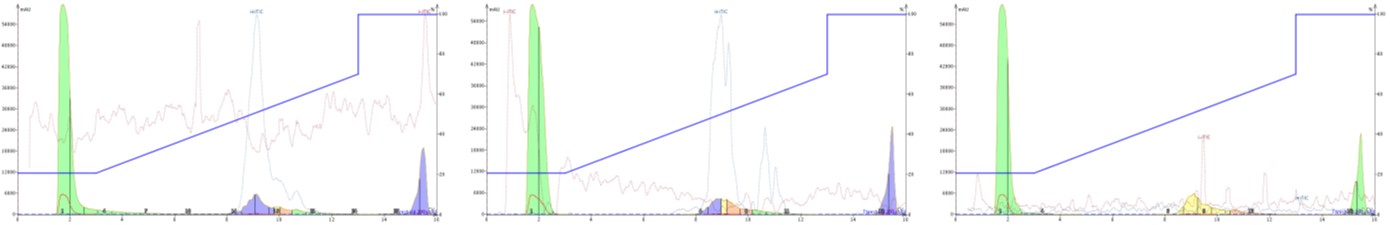
Figure 2. Purification of GLP-1 with an acidified mobile phase and acidified carrier solvent (left), unmodified carrier solvent (middle), or basic carrier solvent (right). The dotted red and blue lines overlayed on the chromatogram represent the negative and positive total ion chromatograms. Positive ion peaks overlay with the UV absorption profile for the peptide only with acidic and unmodified carrier solvents.
There is clearly evidence of mobile phase influence in these results. The unmodified carrier solvent is composed of neat acetonitrile and water, and is not buffered to any extent. Despite this, positively charged ions are still detected and if we take a closer look at the mass spectrum where the peptide elutes, a reasonable signal for each of the identified peptide ions is detected, Figure 3. When compared to the ionization efficiency provided with an acidified carrier solvent, I don't think an unmodified carrier solvent will actually enable sufficient ionization to trigger fraction collection, Figure 3.
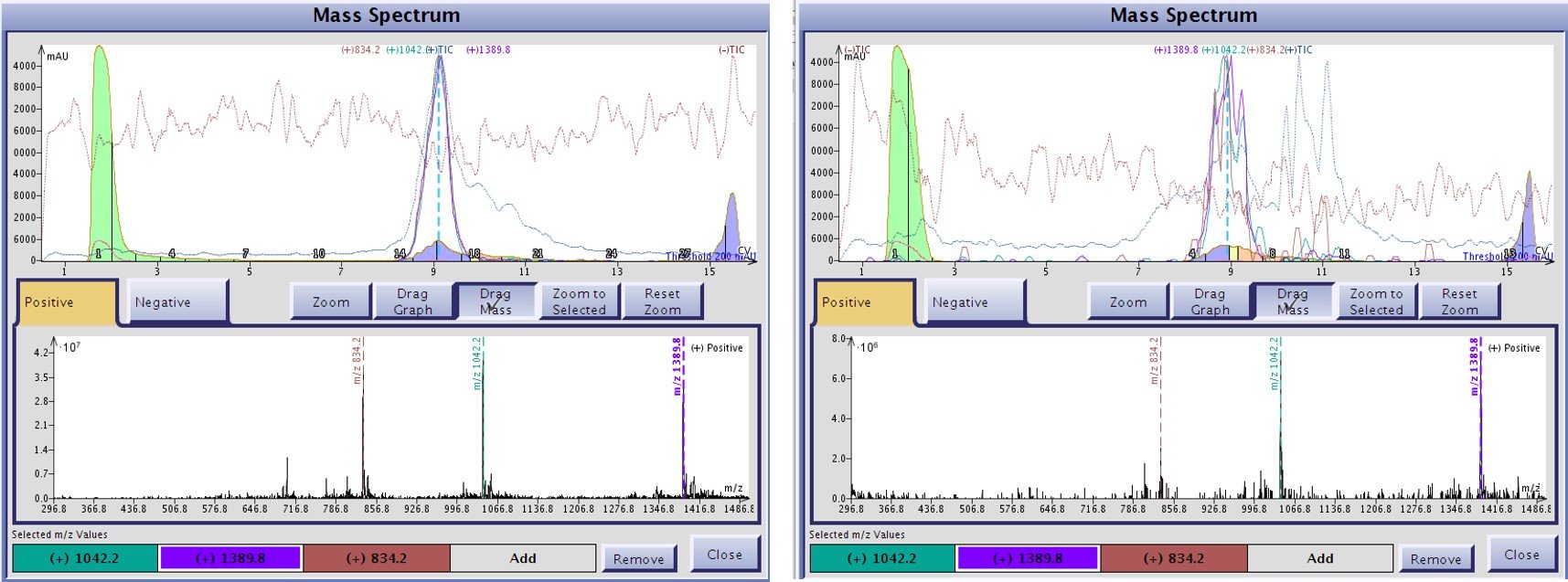
Figure 3. Level of ionization detected for GLP-1 associated ion when using either acidic (left) or unmodified carrier solvents (right). While both carrier solvents enable positively charged ions to be detected, the acidified carrier solvent generates ions more efficiently and increasing the probability that fraction collected can be triggered appropriately.
Sometimes, we change the mobile phase modifier to improve the final purity of the desired peptide. One of the easiest strategies in this regard is to alter the pH from an acidic regime to a basic regime by adding 0.1% ammonium hydroxide to the water and acetonitrile. Given that the GLP-1 peptide is likely to be negatively charged at elevated pH, I decided to look for negatively charged ions as well, Figure 4.
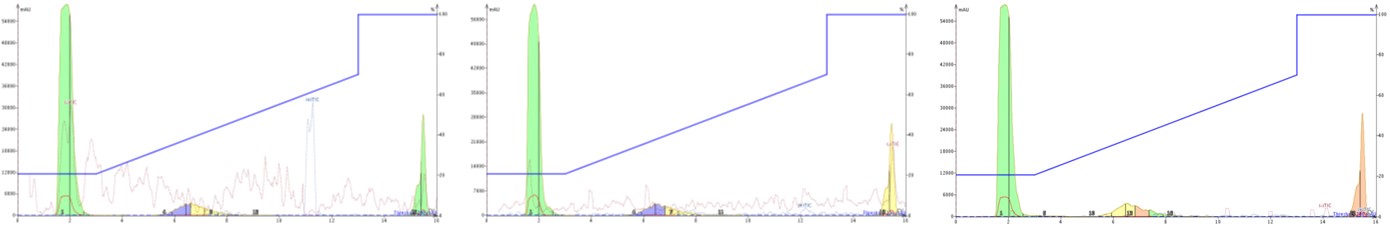
Figure 4. Purification of GLP-1 with an basic mobile phase and acidified carrier solvent (left), unmodified carrier solvent (middle), or basic carrier solvent (right). The dotted red and blue lines overlayed on the chromatogram represent the negative and positive total ion chromatograms. The peptide is not ionized as either a positive or negative ion to any significant extent under any conditions presented here.
First, notice that with basic mobile phase conditions, the peptide elutes earlier and with a slightly broader peak shape. The earlier elution is expected due to the increase in overall polarity of the peptide under basic conditions. Interestingly though, none of the carrier solvent conditions evaluated provided a detectable negatively charge peak in the total ion chromatogram (TIC). I don’t have a good explanation for why a peptide, that should be charged leaving the column and entering the fluidics for the mass spectrometer, isn’t generating any measurable mass spectrum at this point. One thought is that the alternative charge state of the peptide under basic conditions (protonating all the acidic residues) causes a conformational change that in some way shields the remaining charged residues, significantly limiting detection by the mass spectrometer.
Given these results, it is clear that this particular peptide is more readily detected as a positive ion, even with matched basic mobile phase and carrier solvents. And this is likely to hold true for most peptides, particularly longer sequences. In previous work, I observed contradicting results to these, but that was for a much shorter peptide sequence.
To learn more about strategies that enable purification and recovery of highly pure peptide with flash chromatography, follow the link below.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership