You’ve just finished a peptide synthesis and now it’s time to cleave the peptide from the resin. You’ve selected a specific cleavage cocktail, performed the reaction and now what? The vast majority of peptide chemists will precipitate their peptide using an ether solution, lyophilize, and move on to purification. But is that the only option?
In today’s post I’ll highlight an alternative strategy that saves both processing time, potentially dangerous reagents, all without compromising the integrity of the recently synthesized peptide.
Almost every peptide cleavage protocol I have read/identified ends with “collect the peptidyl solution in a cold ether solution and recover the precipitated peptide product” or similar. This step serves as a very crude clean-up of the peptide-containing solution by retaining liberated protecting groups, very short truncation products and sometimes quenching side chain associated reactions in the ether solution while precipitating out the desired peptide and related products.
While there are probably many different specific protocols that have evolved over the years, this is generally the steps we followed in my lab:
-Chill a 10x volume (compared to the cleavage cocktail added to the resin) of 50:50 solution of diethyl ether:petroleum ether on dry ice during the peptide cleavage reaction, or a minimum of 30 minutes.-Note – this was the mix we used. I’ve also used neat diethyl ether and others have reported using methy t-butyl ether or even cyclopeptyl methyl ether
-Collect the cleaved peptide solution in the cold ether solution and return to dry ice for 20-30 min.
-Centrifuge the peptide-containing ether solution for 5 min at 5k rcf.
-Decant the ether solution
-Allow the peptide “pellet” to dry for at least 10 min open in the fume hood
-Resuspend the pellet in 50% MeCN(aq) then freeze and lyophilize
-Purify peptide by reversed-phase HPLC or store dry
While these steps definitely work, they certainly can add significant time to your overall peptide workflow. In my time as an application scientist I have found that I simply don’t have time to wait a couple hours then an overnight lyophilization for many of the experiments I’m working on. So what if there was an alternative strategy? And what are your compromising by pursing an alternative strategy?
The alternative that I have found extremely useful is the Biotage® V-10 Touch Evaporation system, followed by reversed-phase flash chromatography clean up before HPLC polishing (if necessary). The V-10 Touch performs by using extremely high rotation speeds (about 6000 rpms) to create a thin-walled, high surface area cylinder on the inside of a standard scintillation vial. Once the cylinder has formed, vacuum is applied and gentle heating combines to rapidly evaporate the solvent. Importantly, the V-10 Touch is resistant to the corrosive nature of concentrated TFA, allowing me to evaporate completely my cleavage cocktail in a matter of minutes (about 5 minutes for 5 mL of cleavage cocktail – including the water and TIPS).
So now I have essentially a crude peptide film on the inside of the vial, Figure 1. If I know that I’ll be storing the peptide for future purification, I’ll typically give the crude peptide a rinse with 50% MeCN(aq) to ensure that any trapped TFA has been removed, again using the V-10 Touch to evaporate the aqueous solvents in a matter of minutes. After a couple washes, the crude peptide is then stored at -20°C as usual. This process requires about 20 minutes total, depending on how many times I decide to rinse the crude peptide, which is about the time that I would be taking my peptide, in ether, out of the dry ice bucket for centrifugation.

Figure 1. Peptide containing cleavage cocktail before (left) and after (right) evaporation using the Biotage V-10 Touch Evaporation system.
However, most of the time I’m synthesizing my peptides to test a purification hypothesis I might be working on. In this case, I’ll skip the rinsing step all together, measure the approximate weight of the crude peptide recovered, and dissolve the whole sample in DMSO for purification.
So what does this strategy cost you? The short answer is crude purity, Figure 2. The protecting groups are typically not volatile and remain with the crude peptide sample. However, given the significant loading capacity gains observed when using reversed-phase flash chromatography, this becomes much less of a concern. More importantly, the residual protecting groups are typically highly retained by the C18-functionalized stationary phase, essentially eliminating the risk of co-elution with the desired peptide product.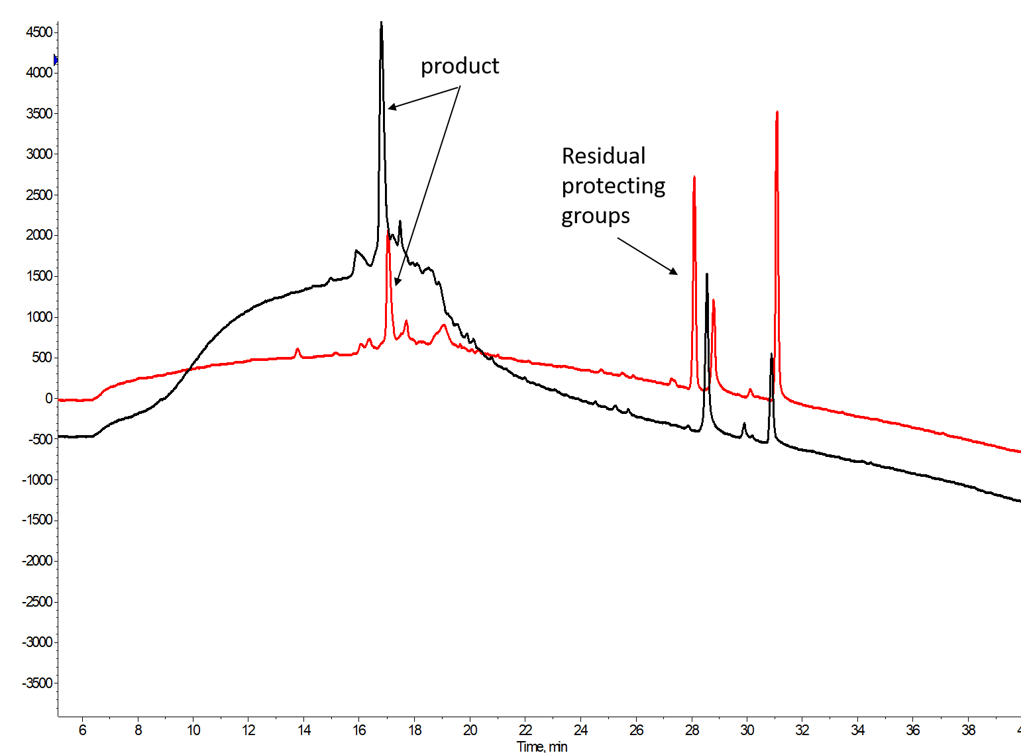
Figure 2. Crude analytical HPLC chromatogram for GLP-1 (1-37) either subjected to ether precipitation (black) or simply evaporated with the Biotage V-10 Touch Evaporation system (red). The only significant difference in the traces is the presence of a single impurity peak containing a compound with molecular weight less than 400 Da.
This strategy eliminates the need for crude peptide lyophilization and allows me to cleave a peptide from resin and purify that peptide in less a single working day, significantly reducing my total peptide workflow time.
To learn more about how Biotage can help improve your peptide workflow efficiency, decreasing the total time to evaluate your purified product, click the link below.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership