Chromatography is as much an art as it is a science. Between synthetic reaction products and natural products, the range of compounds requiring separation, purification, and isolation is broad and diverse creating challenges from time to time. Because of this diversity, not all chromatographic separations can be performed with a “neutral” solvent system – one without added pH modifiers or buffers.
You likely do not know the pKa of the reaction product or by-products so how do you know when to include an acid in your mobile phase? Based on your reaction if the need is separating ionizable/ionic compounds or inherently acidic/basic compounds, mobile phase modifiers can be very beneficial. In this post, I show some examples where lowering mobile phase pH improves either peak shape or improves separation selectivity.
Low pKa compounds
When purifying compounds with low pKa, it is a good idea to acidify the mobile phase to minimize or eliminate compound ionization. An excellent rule of thumb is to lower the mobile phase pH 2 units below the target compound’s pKa. With reversed-phase, this can be accomplished by adding formic, acetic, or trifluoroacetic acid to each solvent or creating a third acidified solvent which can be blended isocratically if your flash system has that capability. The isocratic approach is what I used in the following examples, 1) the separation of hippuric acid (pKa 3.6) and isatoic anhydride with my Biotage® Selekt system and 2) the purification of an amide reaction product using my Biotage® Isolera Dalton 2000.
With example 1, under neutral conditions, we see major fronting of the hippuric acid peak and a distorted isatoic anhydride peak, Figure 1. These phenomena occur because the compounds, especially hippuric acid, are partially dissociated in solution existing as both an intact compound and as an ionized compound. The ionized form is more polar and less retained than the parent compound and thus wants to elute earlier creating the fronting by hippuric acid and the asymmetrical shape of isatoic anydride.
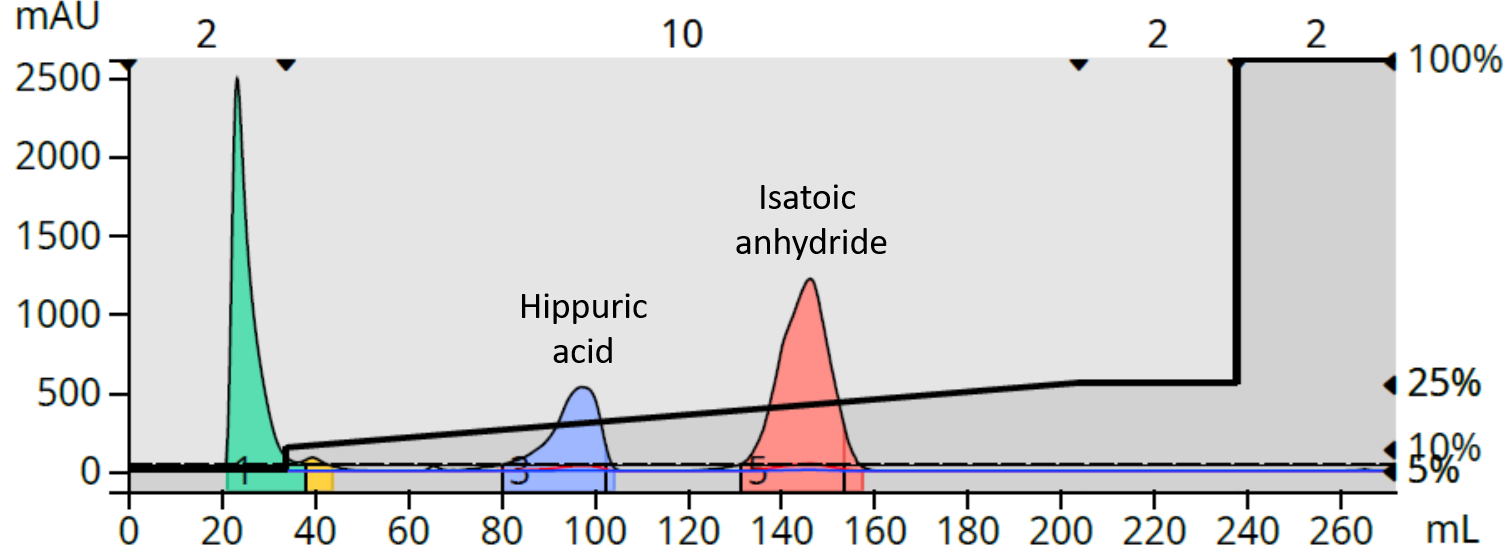
Figure 1. Neutral pH solvent separation of hippuric acid and isatoic anhydride.
To eliminate these issues I added an acidic modifier solvent (1% TFA in water) into the gradient isocratially at 10% of the total solvent composition creating a 0.1% TFA modified mobile phase. The added acid protonated both compounds into their native form sharpening the elution bands and slightly increasing retention (due to their increased hydrophobicity), Figure 2.
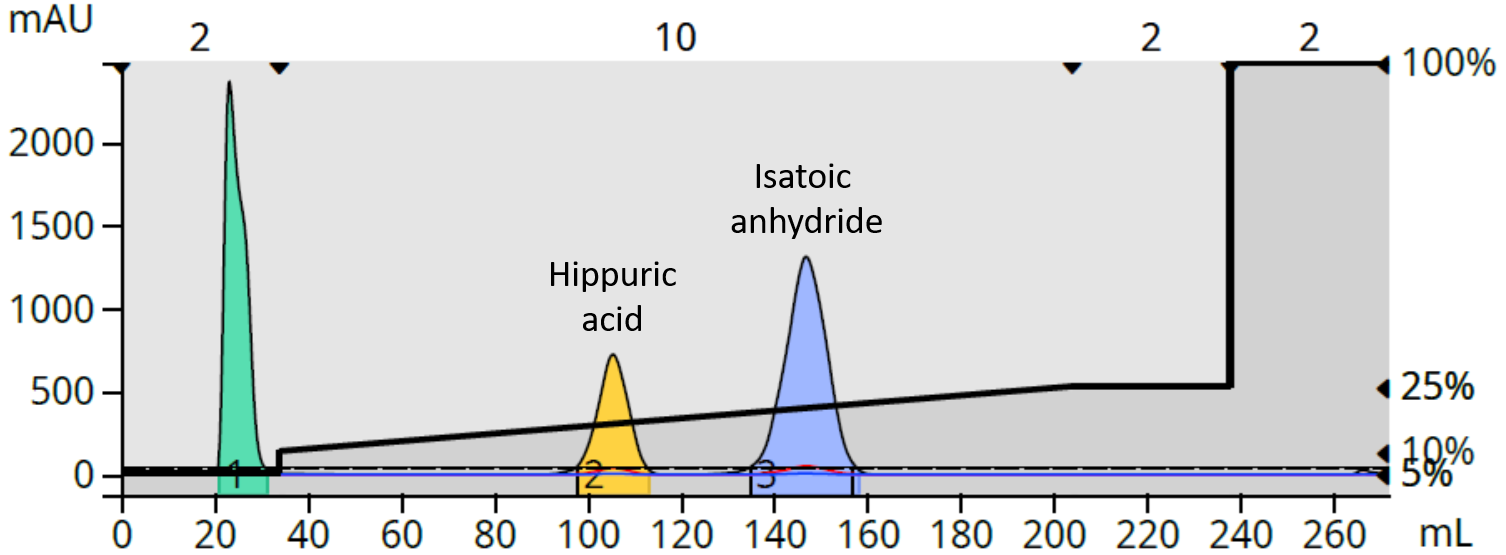
Figure 2. The result of acidifying the mobile phase was a sharpening of both peaks.
Selectivity improvement
A critical aspect of reversed-phase method development is maximizing selectivity, which is the degree of separation between adjacent eluting compounds. Sometimes selectivity can be altered just by changing the organic solvent from acetonitrile to methanol or methanol to acetonitrile. Other times, however, adding acid to the mobile phase impacts chromatographic selectivity by changing the ionization of either the product, the by-products, or potentially all compounds.
We can see this with example 2, the reaction product of succinic acid with α-methylbenzyl amine. Using neutral pH the product (+m/z 221.8) and a by-product (+m/z 342.8) co-elute, Figure 3.
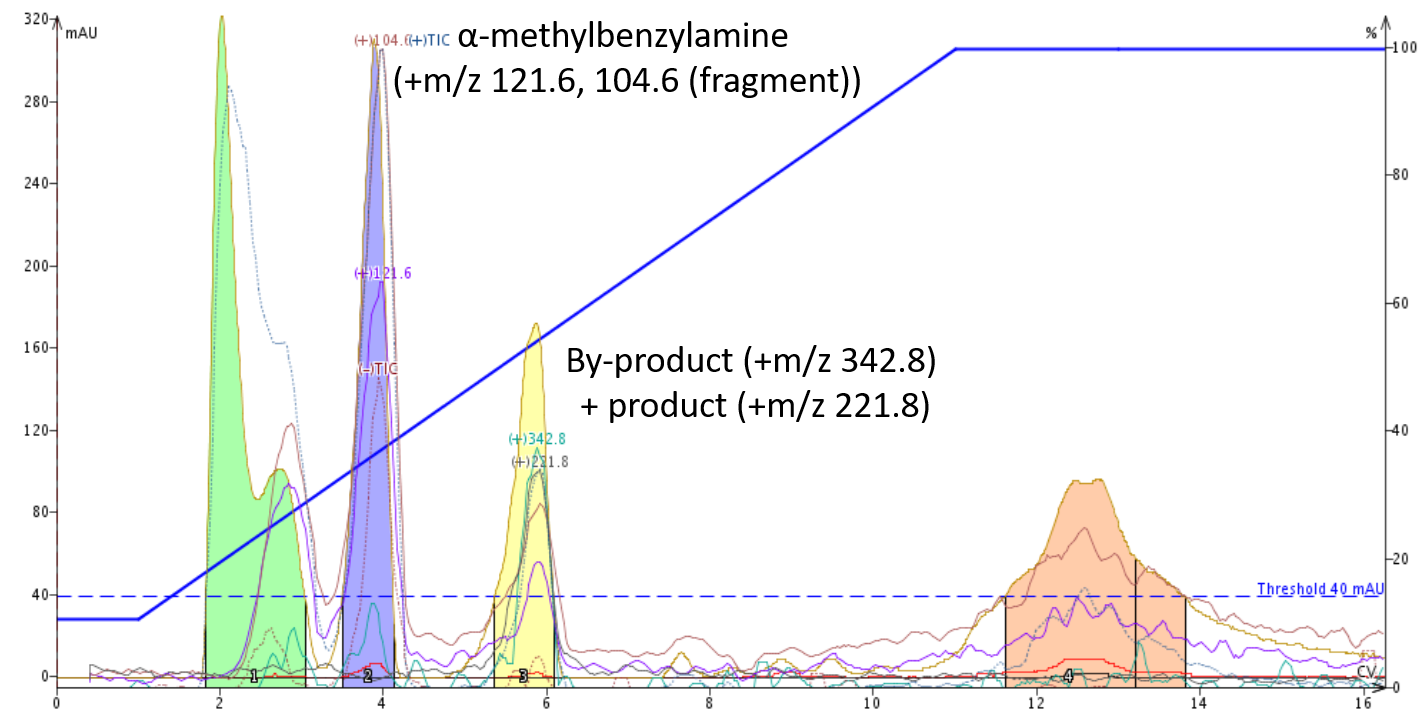
Figure 3. Purification of the amide reaction mixture with a pH neutral mobile phase. The product, +m/z 221.8 has a co-eluting by-product (+m/z 342.8) indicating similar hydrophobicity.
However, by introducing 0.1% acetic acid isocratically as a third solvent, the higher molecular weight by-product was ionized forcing it to elute earlier than the synthetic product while the product retention also increased further improving the purification efficiency, Figure 4.
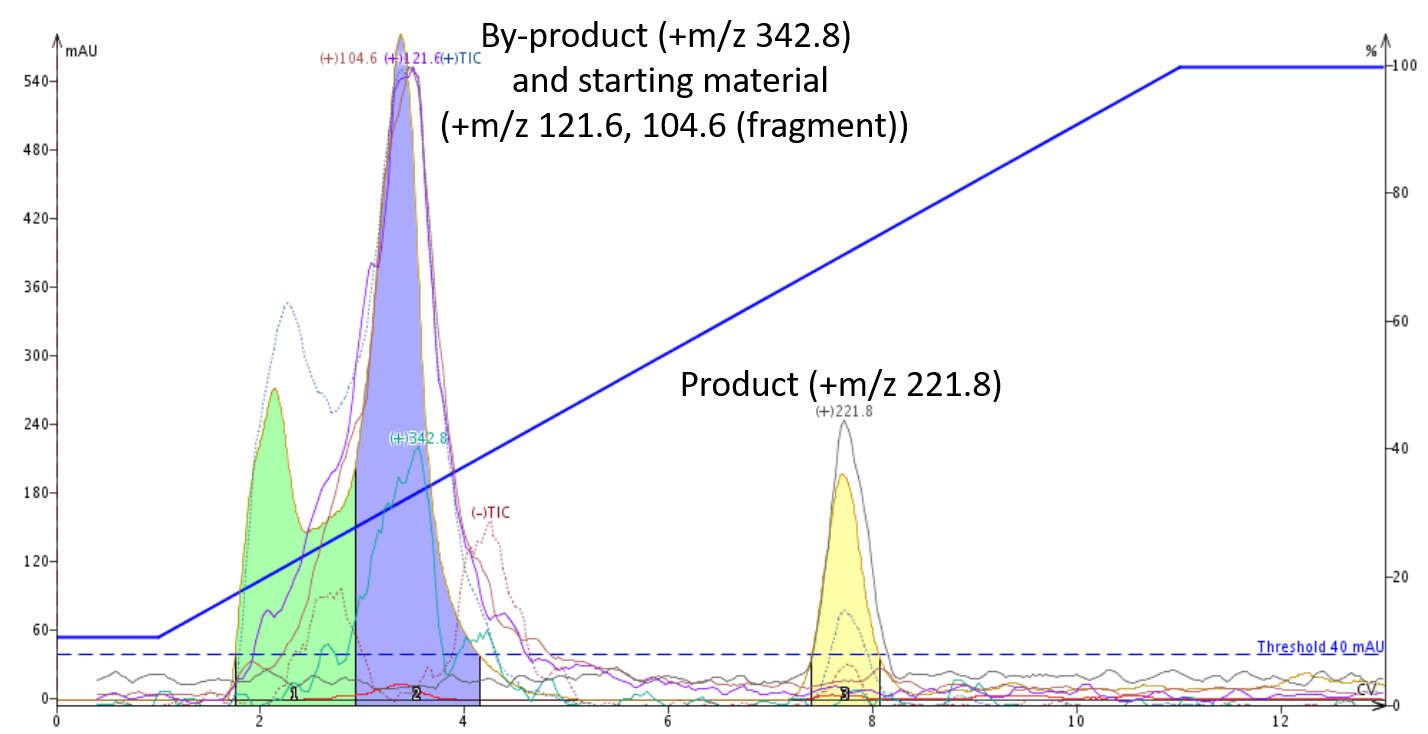
Figure 4. An acidified mobile phase protonated both the product and its co-eluting by-product improving the purification efficiency.
So, what happened to cause both compounds to shift retention relative to each other? Well, by lowering the mobile phase pH a proton is now added to the mobile phase which will cause any compound with a pKa higher than the mobile phase pH to ionize. Ionized compounds are more polar than their neutral version and will have less affinity for the hydrophobic stationary phase and more solubility in the polar mobile phase. Likewise, a compound with a pKa equal to or lower than mobile phase will protonate increasing its hydrophobicity and retention on a reversed-phase column.
Chemistry at work inside a flash column.
More reversed-phase flash chromatography tips are available in my whitepaper, click the link below to download.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership