As you know, reaction chemistry involves determining and selecting the right conditions for optimal product yield and purity. There are actually six variables, that I know of, needing consideration including…
- -Reaction temperature
- -Reaction duration
- -Reaction solvent
- -Reagent concentration
- -Reagent addition order
- -Purification strategy
Previously, I have published posts on the impact of time, temperature, reaction solvent, reagent addition order, and purification strategy. In this post I address how reagent concentration impacts the yield and purity of the synthesis of 2-amino-N-benzylbenzamide, Figure 1.
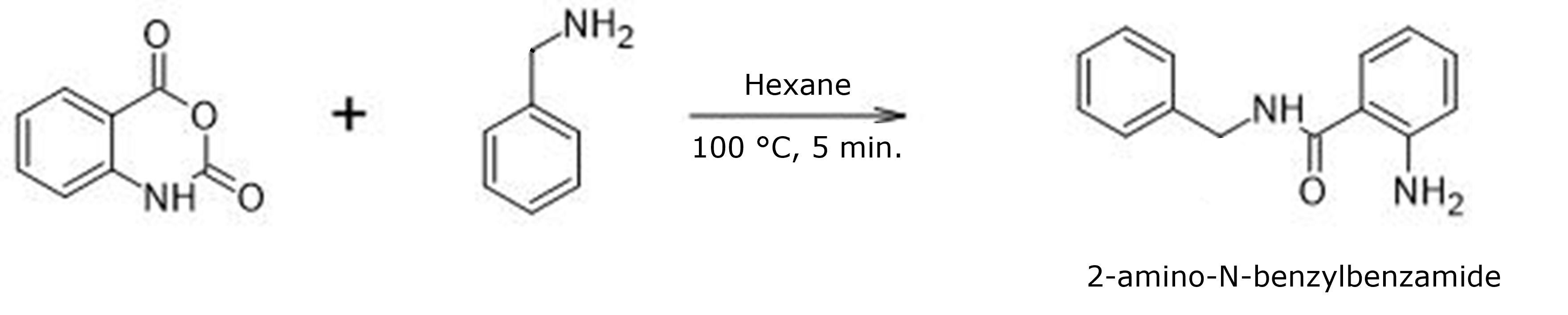
Figure 1. Reaction of isatoic anhydride and benzylamine.
There is nothing particularly difficult about this synthesis but crude product yield and purity are impacted by each of the above synthetic variables, different reagent concentrations included. By increasing the number of reagent molecules in a fixed volume the chances of a chemical interaction or bond forming theoretically increase. While that is true, I wondered if there is an optimal concentration at which crude yield and purity are maximized? If so, how does that impact downstream reaction mixture purification?
So, I performed some experiments to hopefully uncover the answers and to share those results with you. My experiments all used a reagent ratio of 1 to 2 - 1 part isatoic anhydride to 2 parts benzylamine. Reaction conditions were 100 °C for 5 minutes in hexane, with the actual synthesis performed with a Biotage® Initiator+ microwave reactor, Table 1.
Table 1. Reaction scales and crude yields

We can see by the data in Table 1, that actual synthetic yields were greater than the theoretical yields. These differences are due to the creation of by-products which, of course, need to be removed from the actual synthetic product. For this step, normal-phase flash chromatography is the typical go-to technique. Using silica flash columns (10 g, 25 g, and 50 g Biotage® Sfär HC) and a hexanes/ethyl acetate gradient on a Biotage® Selekt flash system, I was able to remove many less-polar and highly polar by-products, Figure 2.
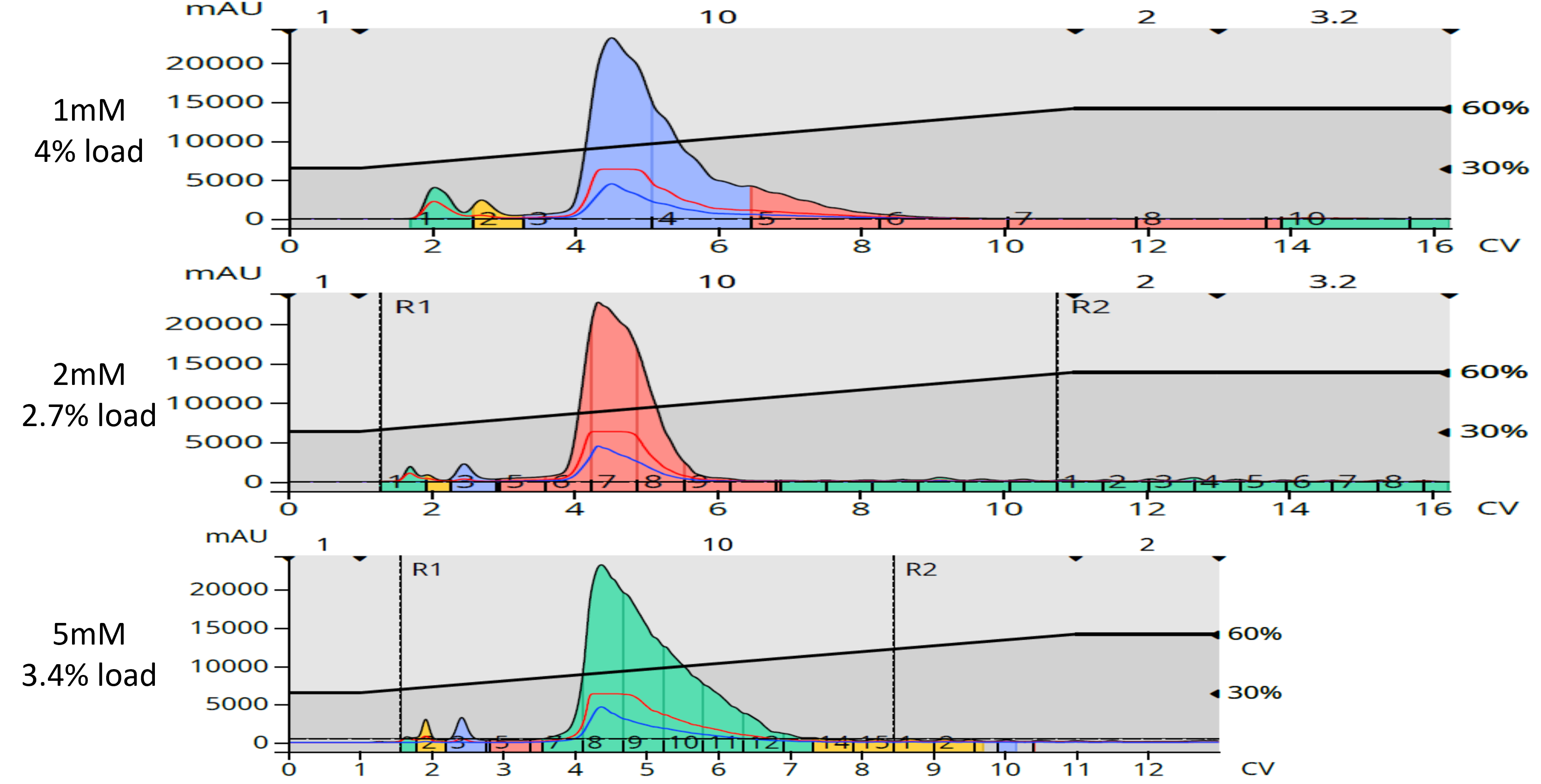
Figure 2. Normal-phase flash purification of the three reaction mixtures show similar results. Less polar by-products eluting early and more polar by-products either eluting late or not eluting at all.
As expected, the normal-phase clean-up of the crude reaction mixtures lowered the total yields significantly, based on the solids recovered after drying the major peaks’ fractions, Table 2.
Table 2. Normal-phase flash chromatography product yields

Interestingly, purified product yields were least with the 1 mM reaction but very similar with the 2mM and 5mM scale reactions – an indication that larger reaction scales do impact product yield. However, the dried purified products were found not to be as pure as desired as each of the three reaction products retained some brown/tan color, likely from co-eluting isatoic anhydride by-products (isatoic anhydride is brown).
To remove this color completely, I re-purified the normal-phase isolated products using reversed-phase flash chromatography (Biotage® Sfär C18) with a water/methanol gradient. This secondary, or orthogonal, purification proved successful not only eliminating the brown color but also removing several previously unknown, co-eluting by-products from normal-phase purification, Figure 3.
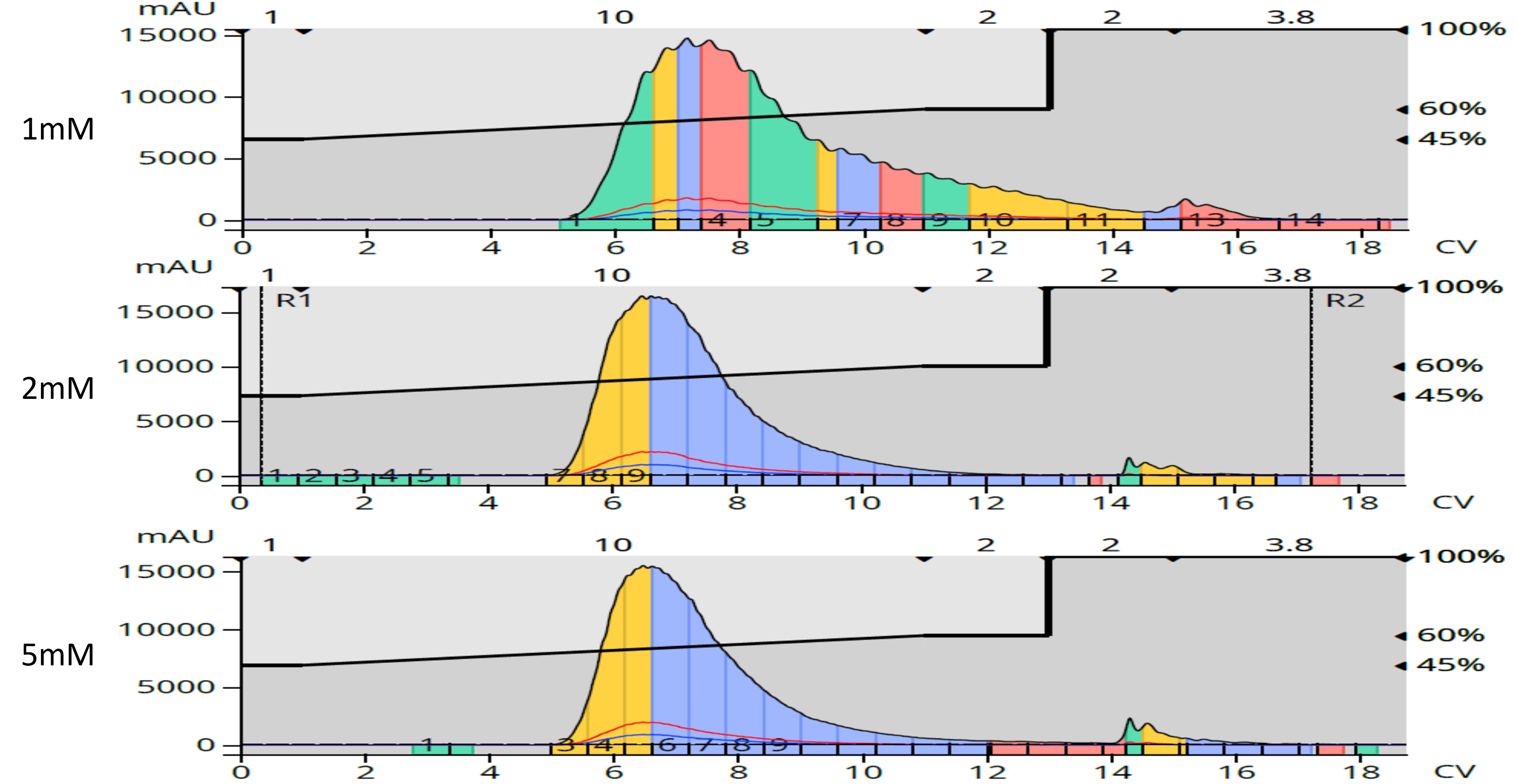
Figure 3. Reversed-phase flash purification of the isolated products from normal-phase removed both polar and non-polar by-products including the compounds contributing to the brown color.
Each of the re-purified product peaks were evaporated and yields determined, Table 3.
Table 3. Reversed-phase flash purification product yield
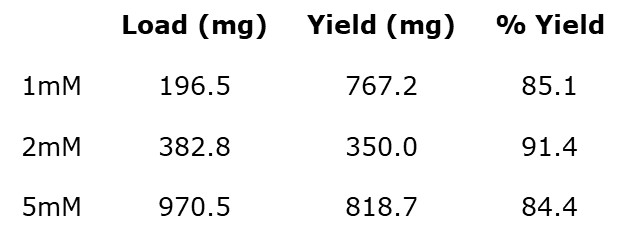
As can be seen with this data, maximum purified yield was obtained at the 2mM scale reaction, which tells me that there is an optimal reagent concentration to maximize both yield and purity for this, and likely other, reactions.
Interested in learning more about microwave synthesis? Download our reaction tutorial.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership