Think of orthogonal flash chromatography as 2D-chromatography where a reaction mixture or natural product extract is purified first with one methodology or solvent gradient then re-purified with a different method or solvent pair in order to remove co-eluting impurities. This is a technique practiced in medicinal chemistry, especially for final compound purification, when the final product is purified first with normal-phase flash followed by reversed-phase prep HPLC.
There are two general flash chromatography techniques...
- -Normal-phase that uses columns packed with a polar media (usually silica) through which non-polar/moderately polar solvents (hexane, ethyl acetate, dichloromethane, etc.) are pumped
- -Reversed-phase, where the column is packed with a lipophilic media (typically C18 bonded silica) and through which polar solvents (water, methanol, acetonitrile) are pumped
Often, one or the other technique is required based on the target compound’s chemistry and those of any by-products. But here is where the chromatography can get tricky or complicated as on many occasions in my experience neither methodology provides totally adequate purity, even with method development. It is these situations where orthogonal flash chromatography (normal-phase followed by reversed-phase, or vice versa) can provide the needed purity levels. This technique can also replace semi-prep HPLC for final cleanup saving time and solvent!
One example that I will share is the purification of an amide I synthesized via microwave (Biotage® Initiator+), Figure 1. One of the starting materials, isatoic anhydride, is brown and that color follows through from the reaction through extraction in DCM and reversed-phase flash chromatography even though the vast majority of the isatoic anhydride is consumed in the reaction. The result is a tan colored product which, ideally, should be white crystals, Figure 2.
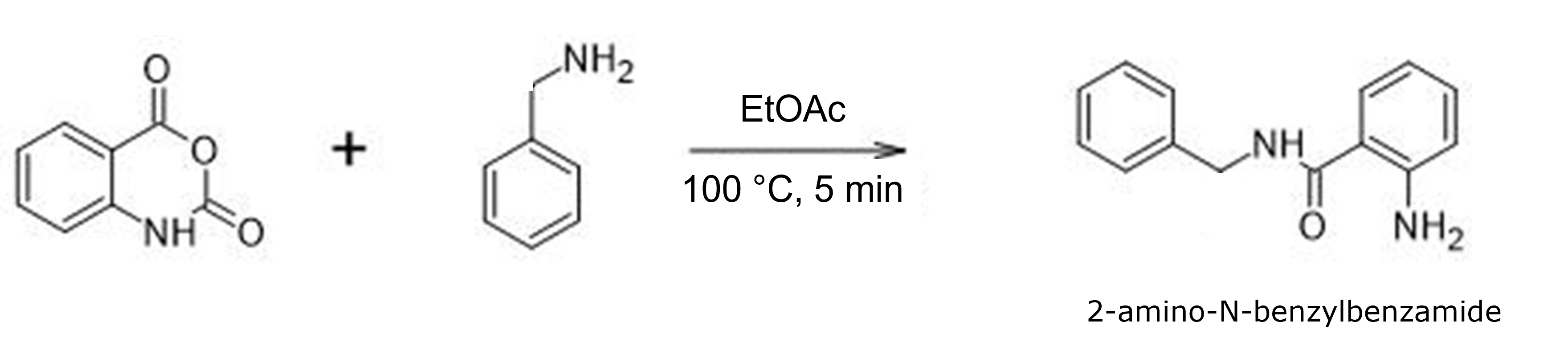
Figure 1. Reaction used in this study, isatoic anhydride + benzylamine in EtOAc at 100 °C for 5 min using MW energy.
%20and%20after%20RP%20flash-1.png?width=3102&name=Orthogonal%20crude%20rxn%20mix%20before%20flash%20(L)%20and%20after%20RP%20flash-1.png) Figure 2. Dried crude reaction mix prior to any purification (left) and after reversed-phase purification. In both cases the brown color from isatoic anhydride is visible.
Figure 2. Dried crude reaction mix prior to any purification (left) and after reversed-phase purification. In both cases the brown color from isatoic anhydride is visible.
Normal-phase TLC with a 80% hexane/20% ethyl acetate solvent system showed the product eluted with an Rf of 0.21 with only two less polar by-products ahead of it with higher Rf values and a very polar impurity stuck at the baseline. Based on this data I determined the purification should be easy with high loading, though I was not sure about the color issue and whether it would elute under these conditions.
Since I already knew that the some of the color carries through with the product using reversed-phase, I decided to try normal-phase flash. I dried the reaction, ~400 mg, on 1.6 g of silica so I could dry load it for purification on a 5-gram Biotage Sfär HC column (8% load).
I decided to use a Biotage® Isolera Dalton 2000 for the purification just in case there were any interfering by-products not separated and detected by TLC. I am glad I did as analysis of the normal-phase results indicated two co-eluting compounds, a by-product (+m/z 315) and un-consumed isatoic anhydride (-m/z 162) in the front and tail of the product peak (+m/z 227), resp., Figure 3. Solvent used – 225 mL, time required – 12.5 minutes.
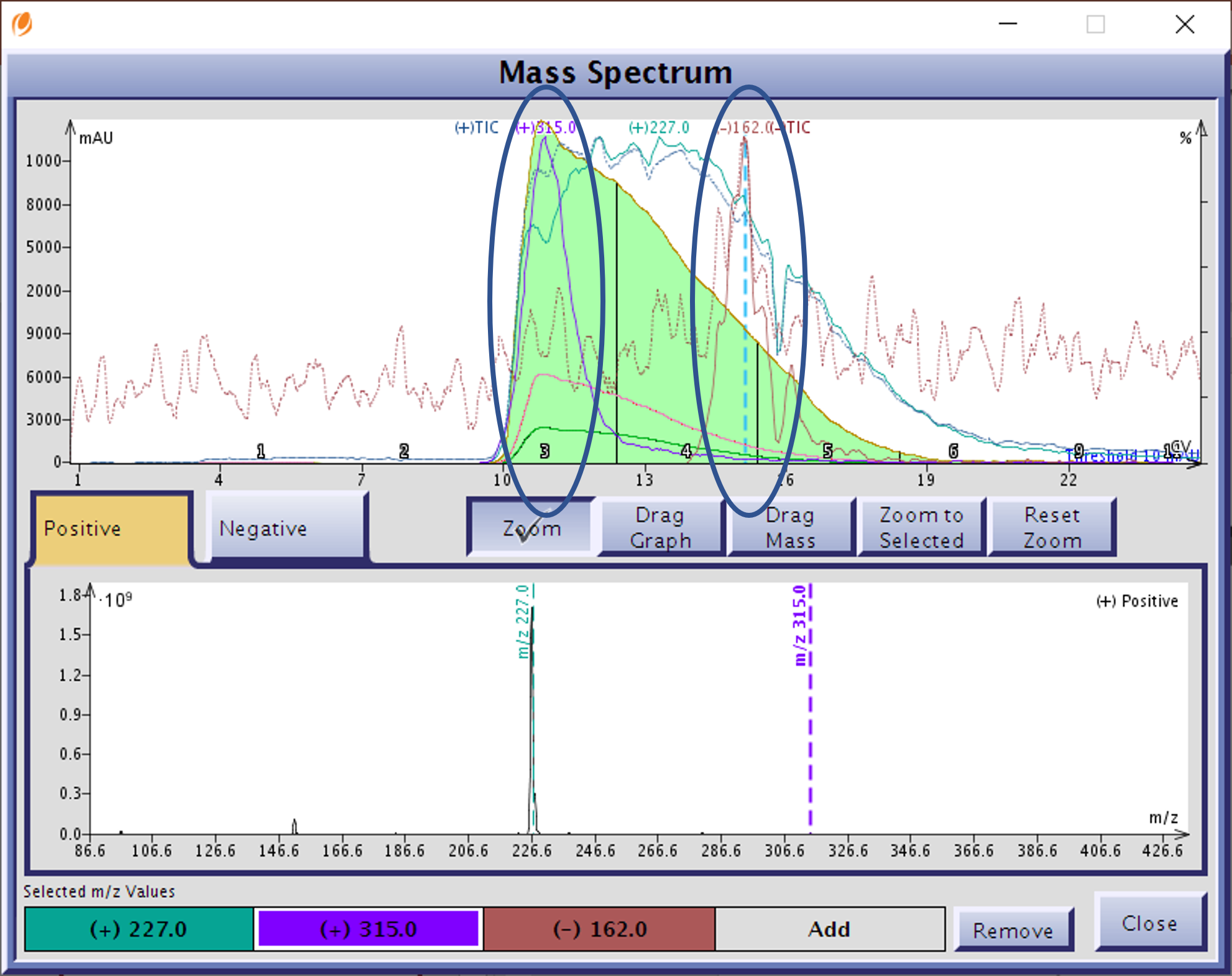 Figure 3. Normal-phase reaction mixture purification showed the two impurities, circled, co-eluting with the product of interest with +m/z 227.
Figure 3. Normal-phase reaction mixture purification showed the two impurities, circled, co-eluting with the product of interest with +m/z 227.
UV spectral analysis also confirmed their existence as there are three different UV spectra depending on where in the peak the spectrum is analyzed, Figure 4. Oh, by the way, the brown color mostly was retained on the dry load silica, an added bonus!
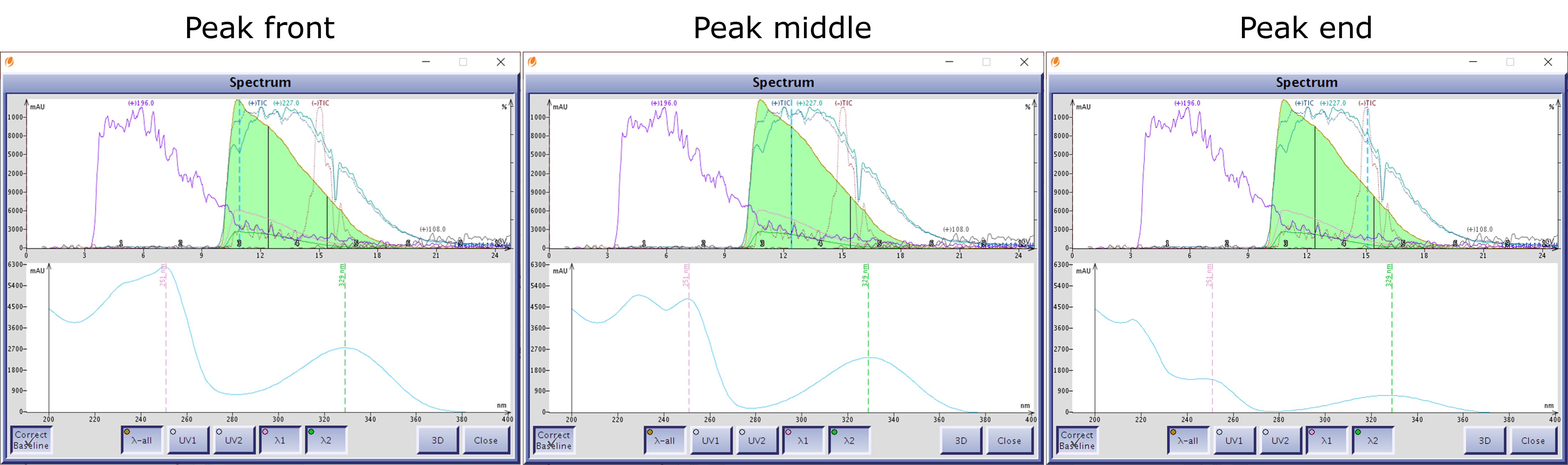
Figure 4. UV spectral analysis of the product peak shows three different spectra depending where it is measured. Left - front of peak. Middle - center of peak. Right - tail of peak.
After evaporating the normal-phase product fractions (Biotage® V-10 Touch), which yielded 302 mg of a light tan solid, I re-dissolved the product in 5-mL of methanol and performed a reversed-phase purification (12-gram Biotage® Sfär C18 column), also using dry loading, this time with 1.2 grams of silica for a load of 2.5%.
The results were a colorless product fraction devoid of both co-eluting impurities, Figure 5. Solvent used – 272 mL, time required – 9 minutes. In contrast, a prep HPLC purification would likely have needed a column of at least 20 x 250 mm packed with 5 μm C18 to achieve this load (quite expensive), would consume 4 to 5 times the amount of solvent, and require an hour to purify!
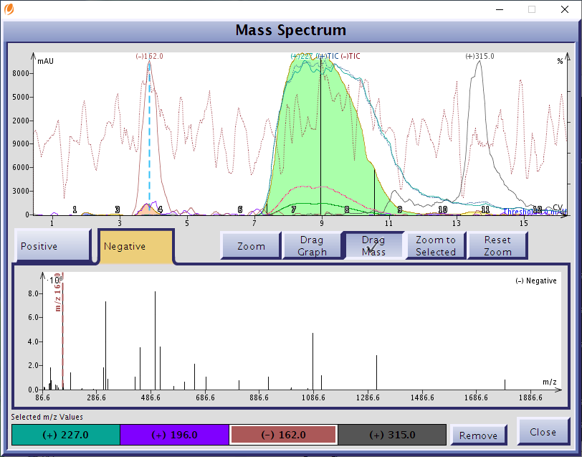
Figure 5. Reversed-phase flash purification removed both major contaminants from the product with the excess isatoic anhydride (-m/z 162) eluting at 4 CV, the by-product (+m/z 315) eluting at 14 CV, and the product (+m/z 227) eluting between 7 and 11 CV.
The + m/z 315 by-product eluted 3 column volumes after the product while the more polar isatoic anhydride starting material eluted 3 CV prior to the product. The evaporated product fractions generated the desired white crystals with a yield of 239 mg (~60% yield from original reaction), Figure 6.

Figure 6. Photo of orthogonally purified product crystals.
So, although normal- and reversed-phase flash are typically used as reaction clean-up steps often requiring further product purification by HPLC, utilizing both normal- and reversed-phase flash will help you achieve the same purify goals faster and with less solvent than prep HPLC.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership