Compounds precipitating during flash chromatography is at best an inconvenience when working up your crude reaction mixture. Precipitation during purification typically happens in the column or in the tubing exiting the column.
In this post, I will propose a strategy that can minimize and perhaps prevent this issue from occurring.
In my 40 year career as a chromatographer, I have both encountered this issue personally and have heard myriad organic synthesis chemists complain of this problem, perhaps you too have had to deal with this.
This problem occurs when one or more compounds in the crude sample have different solubility when being purified by chromatography than they do when they are part of the reaction mixture. During purification, the compounds separate from each other and must remain in solution as the solvent blend becomes increasingly polar (if using normal-phase flash chromatography). Just because your reactants and reaction products may be soluble in the reaction solvent does not mean they all will be soluble in the mobile phase, especially as they become more pure during chromatography.
As mentioned in a previous post, understanding reaction mixture solubility is the first step in optimizing your purification so it makes sense to add a drop of your crude in various solvents to see what “crystalizes” (I love puns! Sorry about that). Once you have determined crude solubility and performed TLC, your method is then transferrable to your flash system. However, the possibility of precipitation still exists as “pure” product may remain soluble during the solubility test (influenced by other compounds in the crude) but when separated are no longer are soluble.
There are two options to eliminate this potential problem:
1. Dry loading, which essentially precipitates all of the crude components on a sorbent allowing them to be selectively solvated and eluted during purification
2. Use a mobile phase modifier. This option I discussed previously in regards to pH adjustment in reversed-phase chromatography, but it can be implemented for normal-phase as well.
Implementing a modifier can be a simple strategy to ensure compound solubility. You can either add a co-solvent to your individual mobile phase solvents or, if using a Biotage® Isolera or Selekt system, make use of its additive or modifier feature, Figure 1. With a third and fourth solvent reservoir available, the Isolera and Selekt provide the ability to pump a third solvent isocratically (constant %) during the gradient while allowing your primary solvent reservoirs to remain unaltered.
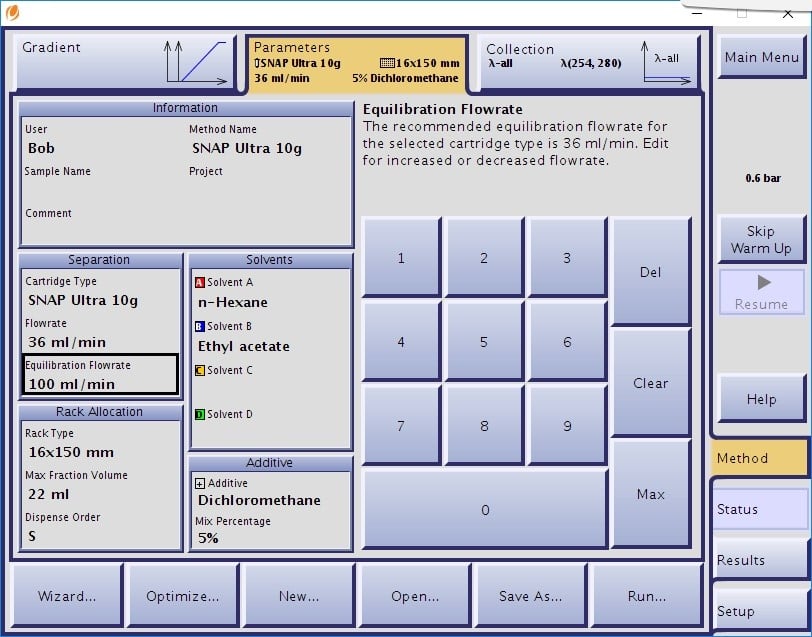 Figure 1. Including a co-solvent additive into a gradient can help prevent compound precipitation during purification. Isolera Spektra and Selekt Spektra systems provide this capability.
Figure 1. Including a co-solvent additive into a gradient can help prevent compound precipitation during purification. Isolera Spektra and Selekt Spektra systems provide this capability.
Incorporating a co-solvent can help prevent untimely crystallization during the run and save you the headache of trying to recover your valuable compound from a packed column or tubing.
How have your dealt with the precipitation issue?
For more flash chromatography tips and tricks, please download our white paper - Successful Flash Chromatography:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership