In my previous post, I talked about the "Chemistry Behind Normal-phase Flash Chromatography", the most common form of liquid-solid chromatography. In this post, I focus on reversed-phase flash chromatography and how it differs from normal-phase.
Normal-phase Chromatography
As a recap, normal-phase chromatography uses a polar stationary phase and a non-polar/moderately polar solvent mobile phase. The separation mechanism is adsorption/desorption with the most polar compounds adsorbing better (and eluting later) than less polar compounds.
Reversed-phase Chromatography
Reversed-phase chromatography is different from normal-phase in three ways...
- -Stationary phase (hydrophobic)
- -Mobile phase (polar solvents - water + methanol or acetonitrile typically)
- -Separation mechanism (partitioning)
What Happens Inside the Column
With reversed-phase chromatography, the chemical mixture (e.g. reaction mix, natural product, extract, etc.) is dissolved in a suitable solvent and injected into the column. When the mixture contacts the non-polar media, let’s say octadecylsilane bonded silica (aka C18 or ODS), the mixture’s compounds will stick to the hydrophobic surface but at differing rates. The more hydrophobic the chemical the more strongly attracted to the reversed-phase media.
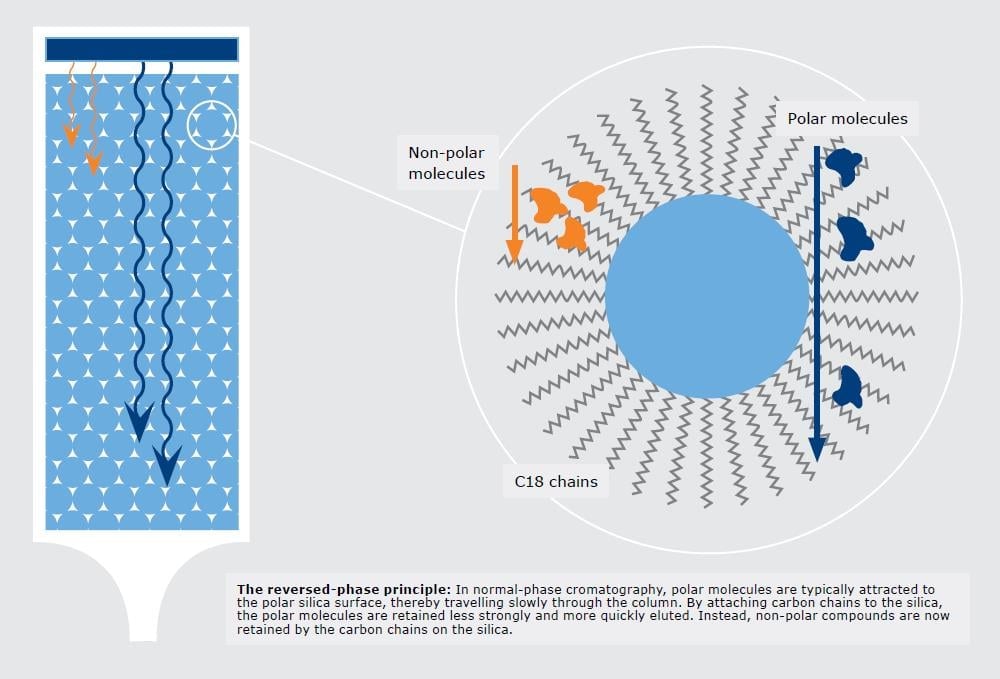
Figure 1. Reversed-phase separation partitioning mechanism binds lipophilic compounds while allowing higher polarity compounds to separate and elute earlier.
With the partitioning mechanism, those molecules with greater hydrophobicity (not necessarily polarity) will adhere to the lipophilic media most strongly which can even separate a double bond from a triple bond, Figure 2.
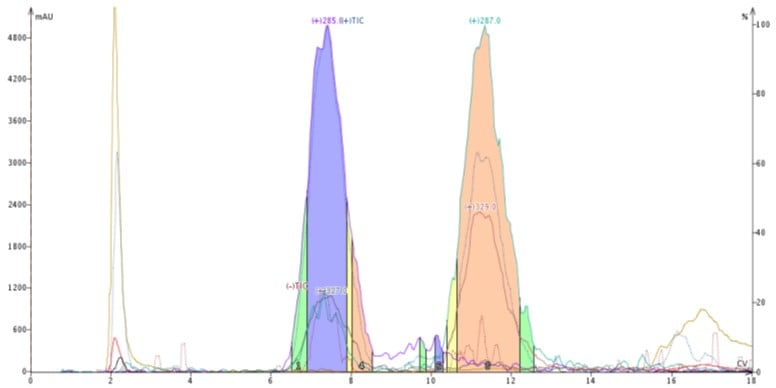
Figure 2. Reversed-phase purification of a natural product extract. The leading compound has a triple bond and the trailing compound has a double bond with all other structural components identical. These compounds are not easily separable by normal-phase chromatography.
To ensure good sample component binding, the sample to be purified needs to either be dissolved and loaded into the column in a chromatographically weak solvent (water, DMSO, DMF) or, if not well solubilized, dissolved with a stronger (non-polar) solvent, then mixed with an inert media (e.g. silica, C18-bonded silica, diatomaceous earth), dried, and packed into a different column placed in-line with the purification column. This sample introduction technique is called dry loading and eliminates many issues encountered when using a liquid load, especially with a sample dissolved in a strong (non-polar) solvent.
After sample load, the mobile phase solvents are introduced. To achieve a separation the sample components need to partition between the stationary phase and mobile phase at different rates. This is achieved using a mobile phase that starts with high polarity and decreases its polarity over time by introducing a solvent that is more organic but water soluble (e.g. methanol, acetonitrile). This changing polarity separation method is known as a gradient.
With a reversed-phase gradient, those compounds with higher solubility in water elute first. As the mobile phase solvent polarity decreases, compounds will begin to partition into the solvent based on solubility in the decreasingly polar mobile phase, Figure 3.
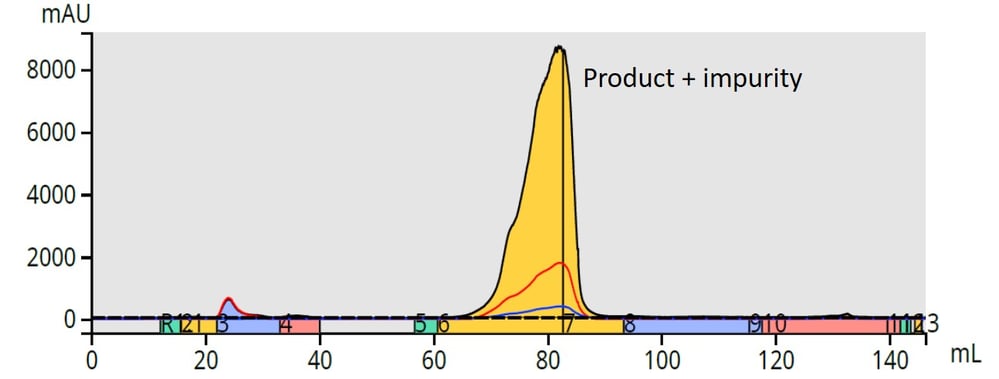

Figure 3. Reversed-phase vs. normal phase of the reaction mixture. While normal-phase flash chromatography (top), failed to separate closely eluting (similarly hydrophobic) by-products, reversed-phase flash chromatography succeeded in separating two less hydrophobic compounds.
So, to summarize reversed-phase chromatography uses differences in compound hydrophobicity, rather than polarity differences, to separate compounds.
Interested in learning more about flash chromatography? Click the link below to download our white paper Inspiring Productivity with Modern Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership