You have performed your synthesis and now it is time to purify the reaction mix. You have used thin-layer chromatography (TLC) and see a separation but when you try to purify with flash column chromatography, you can’t get the target compound separated from an impurity. So, what is happening (or isn’t happening)?
In this post I will give some input on why some separations are not transferable from TLC.
So, what is going on in this scenario? Well, the primary factor influencing chromatographic separations is selectivity so if you are not achieving your separation goal, the separation selectivity must be improved.
Ok, so how do you improve selectivity? By addressing the factors which influence selectivity - solvent choice, media choice, gradient slope. As I have discussed previously, solvent choice is critical for separation optimization, especially with normal-phase silica purifications.
If methanol is used as the polar modifier, then it may be too protic for the flash method even though your compound may be separated by TLC. The reason for this particular problem is that with TLC, the developing solvents migrate at different rates with polar solvents being more retained than the non-polar solvent. With flash column chromatography, this also occurs but since the flash column is typically pre-equilibrated prior to running the gradient (TLC plates are used dry) the actual flash chromatography gradient is different than that seen using TLC.
So, should you then use a non-equilibrated silica column for flash when using a methanolic solvent? Though an option, the amount of heat generated during purification with a non-equilibrated column causes even bigger issues (solvent heating, retention loss, separation loss) so I do not recommend this approach. What I do suggest is looking at an alternative to methanol.
In a previous post I have discussed using acetonitrile as a replacement for methanol. While both are polar, acetonitrile is aprotic which means a TLC method using it will have a better chance of successful transfer to flash than methanol.
To emphasize this point, Figure 1 shows the TLC results of three sample components (a mix of methyl + butyl paraben and 4-methyl-4(5)-nitroimidazole) separated on silica plates with 10% MeOH/DCM and 20% ACN/DCM. Though both TLC solvent systems separate the three compounds only the ACN/DCM solvent blend separates all three components when mixed together using flash chromatography, Figure 2.
 Figure 1. TLC runs comparing separation capabilities between DCM-MeOH (left) and DCM-ACN (right). With each TLC plate, a mix of methyl and butyl paraben were spotted on the left and 4-methyl-4(5)-nitroimidazole spotted on the right. Both elution solvent systems separate all compounds.
Figure 1. TLC runs comparing separation capabilities between DCM-MeOH (left) and DCM-ACN (right). With each TLC plate, a mix of methyl and butyl paraben were spotted on the left and 4-methyl-4(5)-nitroimidazole spotted on the right. Both elution solvent systems separate all compounds.
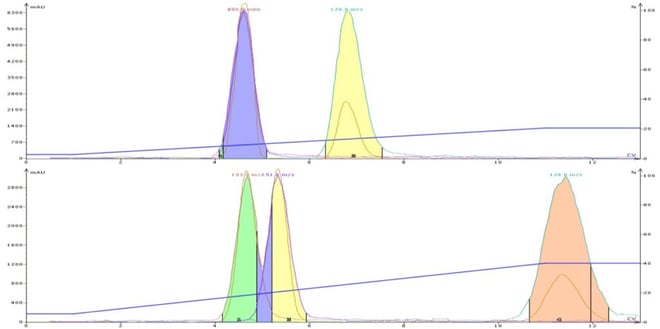 Figure 2. Comparison of a DCM-MeOH separation (top) and DCM-MeCN separation for a 3-component mix of butyl paraben, methyl paraben, and 4-methyl-4(5)-nitroimidazole. The data shows a selectivity and separation improvement with acetonitrile but co-elution when methanol is used.
Figure 2. Comparison of a DCM-MeOH separation (top) and DCM-MeCN separation for a 3-component mix of butyl paraben, methyl paraben, and 4-methyl-4(5)-nitroimidazole. The data shows a selectivity and separation improvement with acetonitrile but co-elution when methanol is used.
So, with the results above in mind, if your TLC method is not reproducible using flash, consider modifying your elution solvents to alter elution selectivity.
Have you experienced a non-transferrable TLC method? Tell us how you solved the challenge.
For more information on flash method development and optimization, please download our white paper - Successful Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership