Normal-phase flash chromatography is an integral component of the organic synthesis workflow. Since reactions rarely generate 100% pure product, they need purification and flash chromatography is the most utilized tool for that task.
But how do you determine the best solvent or solvents to elute and separate your reaction product from by-products and unreacted starting materials? That is this post’s focus.
Determining the best chromatography solvents begins with determining solubility. You need to determine the solvents in which your reaction mixture/product is soluble.
Look at the reaction solvent
Is the crude reaction product soluble in the reaction solvent? If so, this can guide your choice of a component of your mobile phase gradient. For example, if your reaction was run in DCM or EtOAC and remains soluble after cooling, consider a low polarity solvent blend such as hexane with DCM or hexane with ethyl acetate. If your reaction was performed using a polar solvent such as acetonitrile or methanol, you may need to use stronger solvents such as DCM/MeOH, DCM/MeCN, or hexanes/ethyl acetate/IPA (a greener/safer alternative to DCM based mobile phases).
Evaluate solvents using thin-layer chromatography
- Evaluate how well these solvent choices work to separate your reaction mixture using thin-layer chromatography (TLC). Since TLC can be performed in parallel, it is easy and time-efficient to evaluate different solvents and solvent blends with your reaction mixture at the same time. Evaluate the separation results and choose the solvent blend providing the best separation between your targeted compound and its nearest eluting by-products/impurities, Figure 1.

Figure 1. Solvent choice impact on TLC separation of a 5-component sample. Left - 20% EtOAc/hexane. Middle – 100% toluene. Right – 100% DCM.
- Once the solvent blend is selected, repeat the TLC analysis but this time vary the solvent ratios. This test will help you find the most appropriate blend to maximize retention, selectivity, load, and purity, Figure 2.
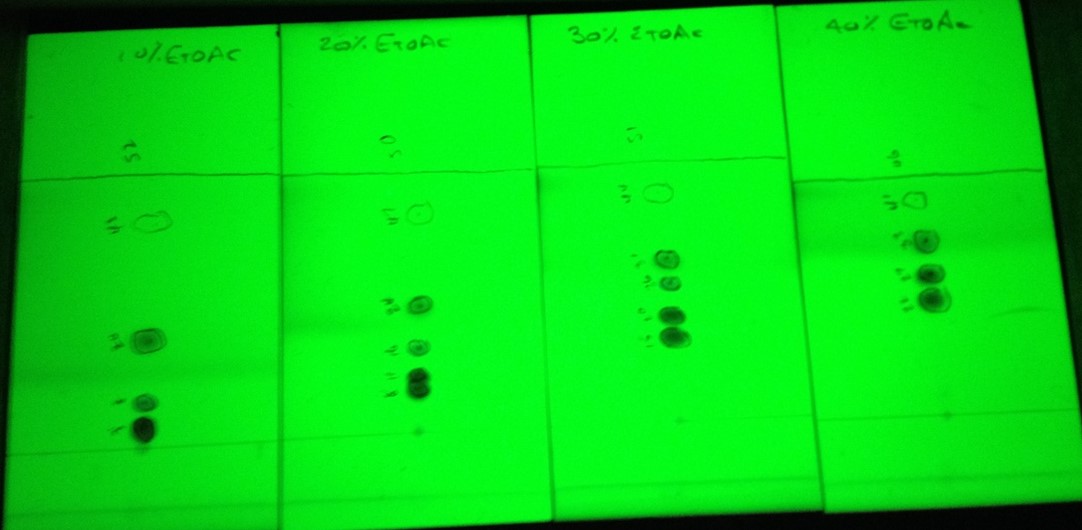 Figure 2. Solvent ratio impact on TLC separation. From left - 10% EtOAc/hexane, 20% EtOAc, 30% EtOAc, 40% EtOAc. For this sample, the 20% and 30% EtOAc data show separation of all 5 compounds.
Figure 2. Solvent ratio impact on TLC separation. From left - 10% EtOAc/hexane, 20% EtOAc, 30% EtOAc, 40% EtOAc. For this sample, the 20% and 30% EtOAc data show separation of all 5 compounds.
Think green
As mentioned above, if you find a DCM-based mobile phase provides the best separation you should consider replacing it with a safer, greener solvent blend of hexane or heptane with a 3:1 ratio of EtOAc + IPA (or EtOH). It has been my experience that this solvent system provides chemists with more flexibility and better purifications than DCM-based mobile phases, Figure 3.
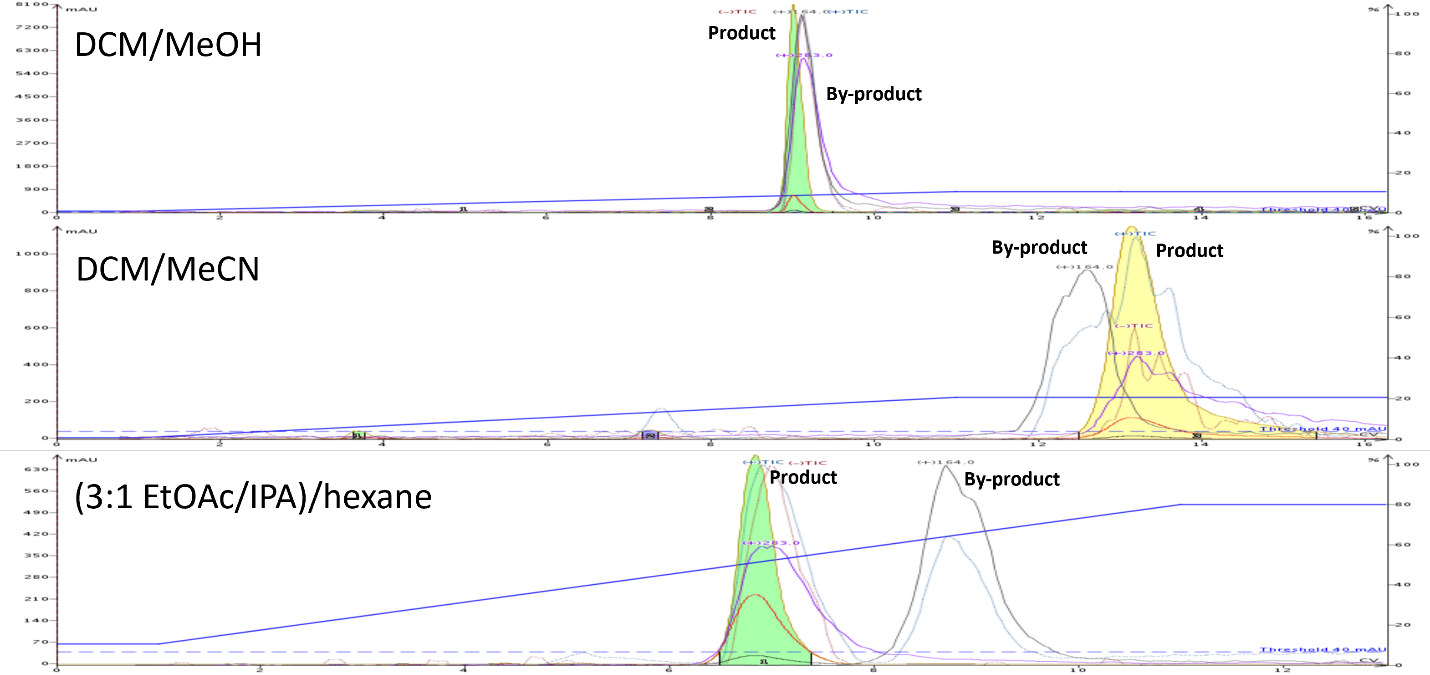 Figure 3. Flash chromatography comparison of 0-10% DCM/MeOH, 0-20% DCM/MeCN, and 10-80% hexane/(3:1) EtOAc + IPA shows a vastly improved separation of the synthetic product and by-product using the hexane/EtOAc/IPA gradient.
Figure 3. Flash chromatography comparison of 0-10% DCM/MeOH, 0-20% DCM/MeCN, and 10-80% hexane/(3:1) EtOAc + IPA shows a vastly improved separation of the synthetic product and by-product using the hexane/EtOAc/IPA gradient.
Determine your elution mode
There are three elution options for your purification…
- Isocratic elution
- Linear gradient elution
- Step gradient elution
Isocratic elution
Isocratic elution is the simplest of the methods as it uses the same solvent ratio used with your TLC. Isocratic elution works best with simple separations, especially when your target compound has a retardation factor (Rf) between 0.15 and 0.4, though load size may be compromised due to significant band spreading, Figure 4.
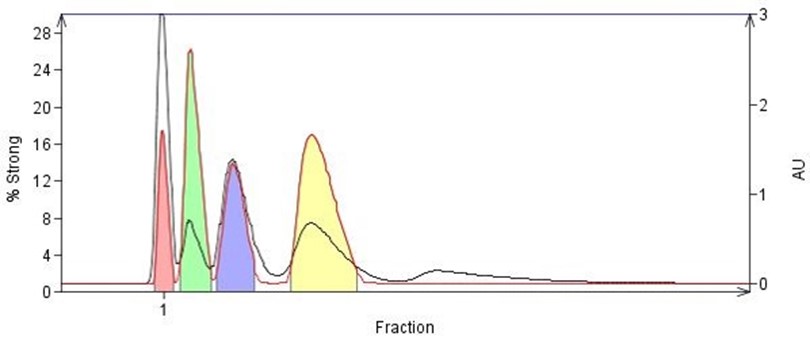 Figure 4. Isocratic flash chromatography based on 20% EtOAc/hexane TLC. The higher Rf value compounds elute quickly while the lower Rf value compounds tend to broaden which can hamper detection as seen here with the fifth eluting compound.
Figure 4. Isocratic flash chromatography based on 20% EtOAc/hexane TLC. The higher Rf value compounds elute quickly while the lower Rf value compounds tend to broaden which can hamper detection as seen here with the fifth eluting compound.
Linear gradient elution
With a linear gradient the elution method begins at a solvent ratio less than that used with your TLC and ends with a ratio higher than the TLC. A common linear gradient starts with a ratio ¼ of the TLC and ends at 2x the TLC ratio over a distance of 10 column volumes (CV), Figure 5.
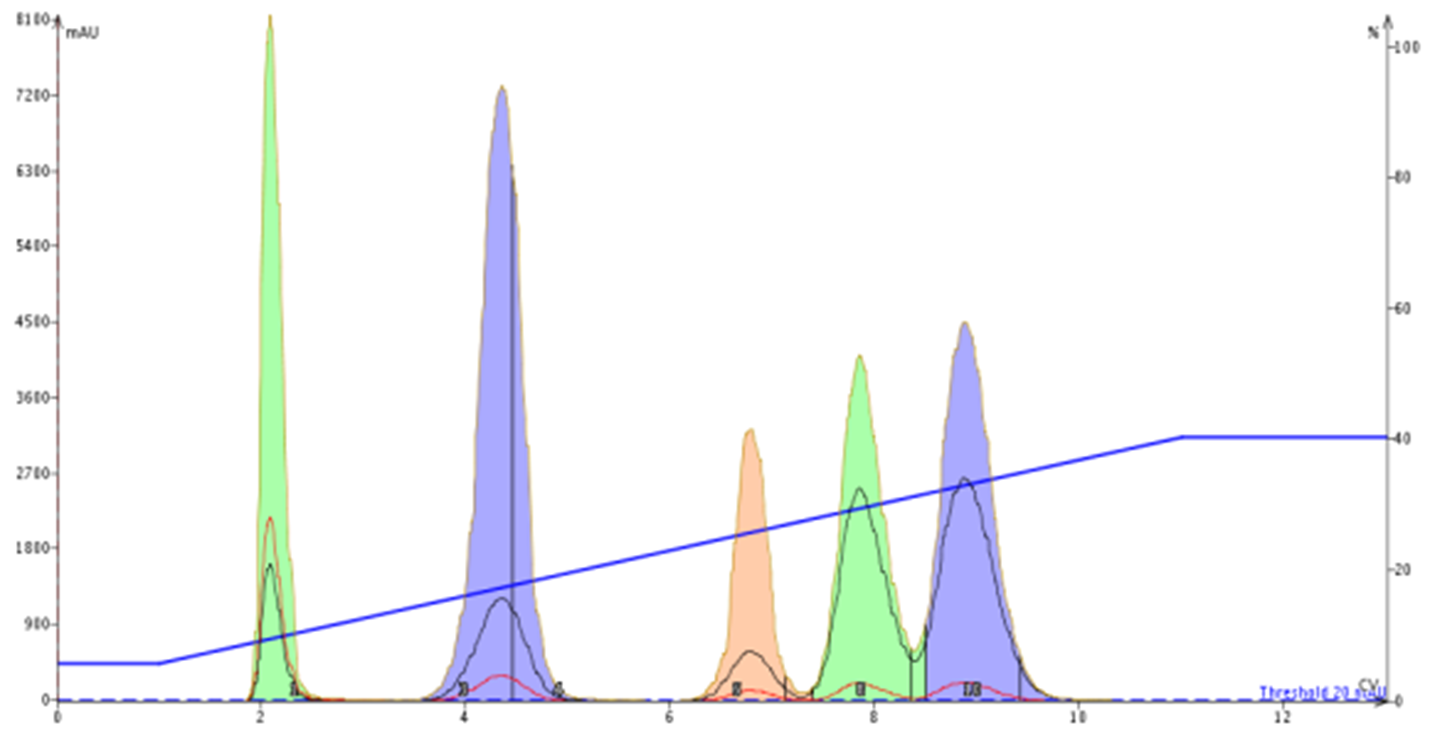 Figure 5. Linear gradient based on 20% EtOAc/hexane TLC for the 5-component mixture increases compound separation and narrows peak widths vs. isocratic elution.
Figure 5. Linear gradient based on 20% EtOAc/hexane TLC for the 5-component mixture increases compound separation and narrows peak widths vs. isocratic elution.
Step gradient elution
A step gradient incorporates a series of isocratic elution steps. These can have steps of different heights and lengths. This purification technique can provide major benefits over both isocratic and linear methods as step gradients can reduce solvent use, help better separate your target compound from its nearest eluting by-products, and sometimes increase sample load. Step gradients are best created by using data from two TLC plates run with the same solvents but at different ratios. Flash systems such as the Biotage® Isolera and Selekt have built-in TLC to step and linear gradient algorithms, Figure 6.
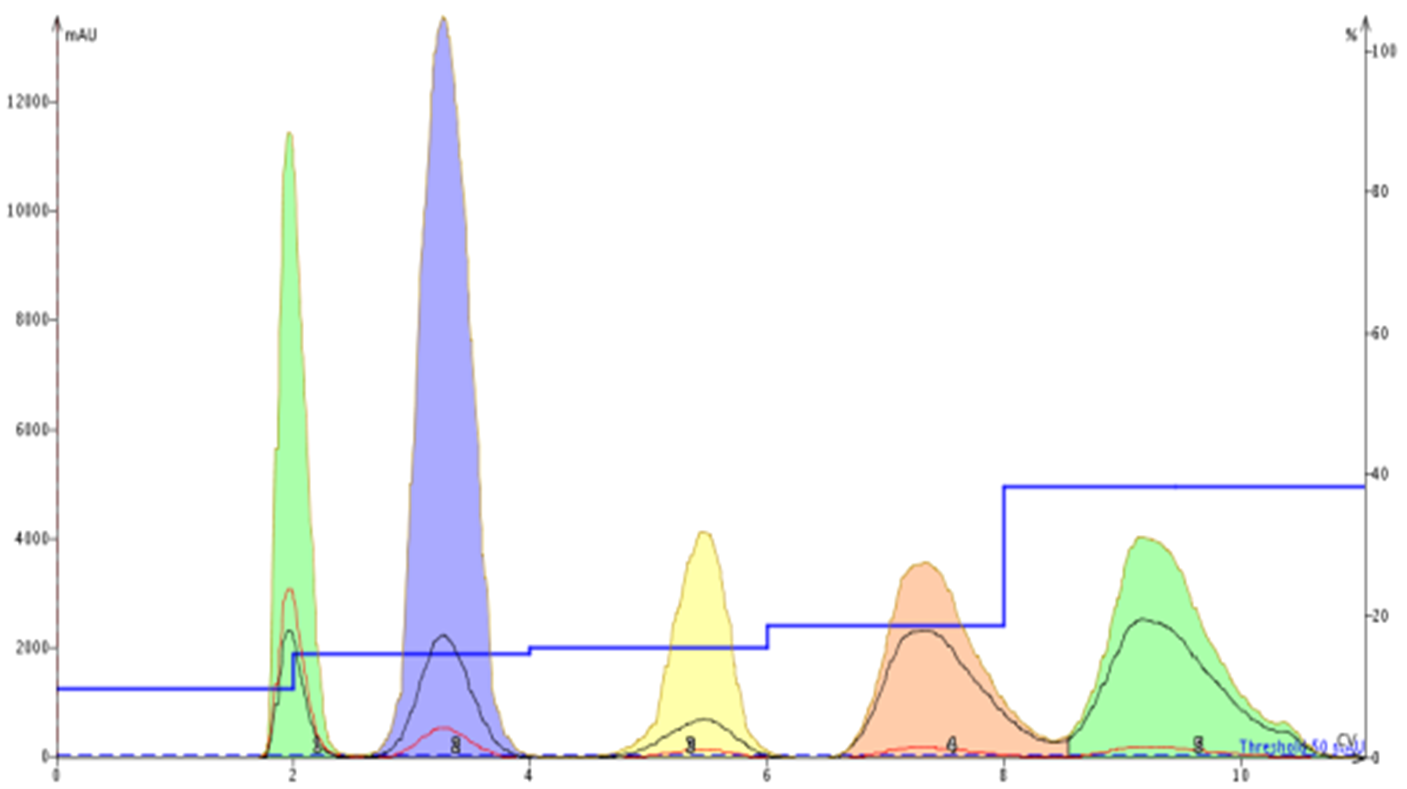 Figure 6. Step gradient based on 20% and 30% EtOAc/hexane TLC data provided a complete separation of each compound with 15% less solvent than the linear gradient.
Figure 6. Step gradient based on 20% and 30% EtOAc/hexane TLC data provided a complete separation of each compound with 15% less solvent than the linear gradient.
So, when creating a normal-phase purification method, be sure to evaluate many solvents as your product purity depends on your solvent choices.
More flash chromatography help can be found in our whitepaper, Successful Flash Chromatography.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership
