I am always grateful for the feedback I get from my blog readers. Today's blog is in response for multiple requests for tips on purifying complex mixtures and suggestions for alternative sample loading techniques.
In this post, I will attempt to address both, to some degree anyway, with a single example using a scavenger resin.
For organic and medicinal chemists synthesizing novel compounds for the first time, reaction by-products and excess starting materials are virtually guaranteed to be present at the end of the reaction. While flash chromatography can often purify the main product quickly with sufficient purity and yield, by-products and/or excess starting materials can interfere with purification success either by partially co-eluting with the synthetic product or by reducing loading capacity.
A good, but underutilized, technique to improve some of the potential interferences is compound scavenging. Scavenging uses a functionalized media to remove unwanted by-products and excess reagents from the reaction mix in order to simplify the purification. This can be accomplished off-line with liquid-liquid extractions and potentially even solid-phase extraction (SPE). But I find those techniques take longer than I like, are a bit laborious, and increase opportunities for sample loss and human errors. The better alternative, in my opinion, is dry-loading my crude sample using a scavenger media as the sorbent.
To illustrate the issue above, I ran a simple microwave-assisted organic synthesis (MAOS) reaction with a Biotage® Initiator+. The reagents used included isatoic anhydride, benzyl amine, benzaldehyde, and glacial acetic acid all dissolved in 3-mL of N,N-dimethylformamide (DMF). The desired product from this reaction is 3-benzyl-2-phenyl-2,3-dihydro-4(1H)-quinazolinone, Figure 1.
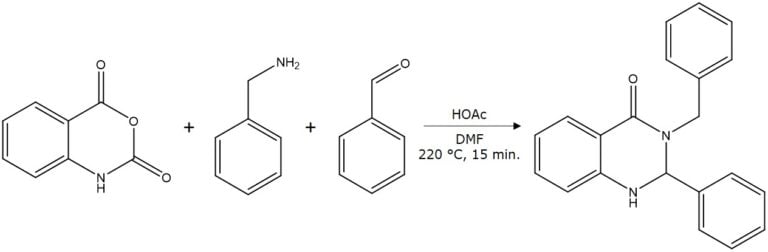
Figure 1. The microwave reaction of isatoic anhydride, benzyl amine, benzaldehyde, and acetic acid is designed to yield the product 3-benzyl-2-phenyl-2,3-dihydro-4(1H)-quinazolinone.
This reaction seems simple enough. According to the literature I followed the reaction, and subsequent purification using silica flash chromatography, should provide >97% purity and a 70% yield. Hmm, 70% yield, where are the losses?
After running the reaction, I ran thin layer chromatography (TLC) analysis of the starting materials and the reaction mixture to see what the other 30% might be, Figure 2. Well, I saw that this reaction created many different compounds, most with high polarity which explains the 30% loss. Great information, but which of these spots is my product?

Figure 2. Reaction mixture TLC analysis shows the presence of many compounds.
I decided to analyze the reaction mix further to see if my synthesis was successful and see what other compounds might be present. I know that my product should have a MW of 314. To do so, I diluted the mix in methylene chloride and injected 50 µL into my flash-MS system (a Biotage® Isolera Dalton 2000). What it detected were strong abundances in positive ionization mode of +m/z 315, 177, 253, among a few others, including 196. This data tells me I have my synthetic product (315, as the M+H ion) as well as several other by-products, Figure 3.
Figure 3. Mass analysis (positive ionization mode) of the MW reaction mixture shows many compounds including the product (315) and several by-products.
To see if the other masses were unreacted starting materials such as benzyl amine, benzaldehyde, or isatoic anhydride potentially detected as adducts or fragments, I used my adduct and fragment finder. This tool did not find any possible adducts or fragments that were logical and associated with unreacted starting materials, so the detected masses are real compounds.
Because the reaction proceeded in DMF, dry loading was used in order to get the best separation (DMF is typically too polar to be used as a normal-phase sample solvent). I used silica as my dry load sorbent and an empty Samplet® cartridge as my dry load vessel, Figure 4. For those unfamiliar with Samplet cartridges they are a convenient way of dry loading and insert directly into the top of Biotage SNAP columns, Figure 5.
As for the process, I mixed 1 gram of silica with 1 mL of reaction mixture (~ 0.25 grams) and dried it using a Biotage® V-10 Touch evaporation system. This sample to sorbent ratio of 1:4 is ideal for dry loading.

Figure 4. A self-packed Samplet cartridge.
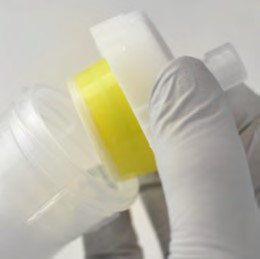
Figure 5. Samplet being inserted into a SNAP column.
So, based on my TLC data and direct mass analysis results, I created a purification method targeting the selected masses +315, +253, +177, and +196. Using a 10 gram Biotage® SNAP Ultra column I ran a 25-100% ethyl acetate in hexanes gradient. The method provided a separation of all four targeted compounds (315, 253, 177, and 196) and several others, Figure 6.
 Figure 6. Crude reaction mixture purification using silica as the dry load media. My reaction product (green peak, fraction 1) has a +m/z of 315 but is contaminated with other compounds with +m/z of 268 and 277 (the red UV trace (254 nm) also shows a trailing shoulder).
Figure 6. Crude reaction mixture purification using silica as the dry load media. My reaction product (green peak, fraction 1) has a +m/z of 315 but is contaminated with other compounds with +m/z of 268 and 277 (the red UV trace (254 nm) also shows a trailing shoulder).
The first peak detected at 2 column volumes (CV) did not have enough response to be collected and had a MW of +m/z 196. I determined that this is the Schiff base product of benzyl amine and benzaldehyde. The first peak collected (my product in green) had a +m/z of 315 (also detected in -m/z as 313) but was contaminated with some co-eluting by-products with +m/z 268 and 277, resp. Fraction 2 also contains product and four by-product masses (+m/z 268, 277, and 253) as well as -m/z 251. It is -m/z that provides the negative ionization signal (red dotted line).
Later to elute were by-products with +m/z 253 and +m/z 177 as well as a few other compounds with positive ionization (278, and 191). Hmm, based on this information I am sure there is more than a 30% yield loss. So, I wondered - how can I improve my product's purity without re-optimizing my flash gradient?
Well, I assumed some of these by-products/impurities were basic (the reaction mixture sure smelled like amines, phew). So, I replaced my silica dry load media with a base-scavenging media, Biotage ISOLUTE® SCX-2 (a propyl sulfonic acid bonded silica ion exchange sorbent). I used the same protocol as with silica (1 mL of reaction mix with ~1 gram of SCX-2), dried it, loaded the media into an empty Samplet cartridge, and performed the same gradient using another 10 gram SNAP Ultra silica cartridge. The results supported my theory that many of the impurities/by-products were basic since most of them were removed, including the large impurity with +m/z 253 (blue peak in Figure 6) and my product's major co-eluting impurity (+m/z 227), Figure 7, a remarkable improvenment.
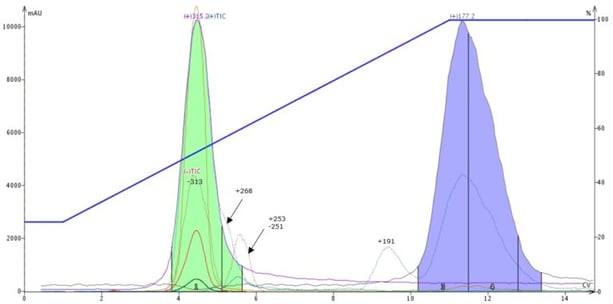
Figure 7. MW reaction mixture purified using a strong cation exchange, base-scavenging media as a dry load sorbent. Most impurities have been removed improving compound purity.
TLC fraction analysis shows my in-line scavenging approach worked. There were no detectable impurities for fraction 1 (the product and major impurity fluoresce as a blue spot making it easy to see) when compared to the silica dry load analysis, Figure 8.
Well, after purification using the dry load scavenger method, my product is 100% pure (by TLC) but my yield was only 31% - this is why I took up chromatography instead of organic synthesis!

Figure 8. Fraction purity analysis using TLC. Left - SCX-scavenged sample. The product is found in fraction 1 and is pure by TLC. Right - Silica sorbent, though acidic, does not scavenge any basic by-products.
Scavenger media is not limited to SCX. Other ion exchange media including strong anion exchange (SAX), weak anion exchange (WAX), and weak cation exchange (WCX) are useful for scavenging and even for a technique called catch-and-release, a topic for future discussion.
For more information on using ion exchange media for sample loading click below:

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership