Gradient or isocratic elution, that is the question. Within flash chromatography these are the options afforded to separate reaction mixtures and natural product extracts and is the focus of this post.
For those unfamiliar with the terminology, isocratic elution means that the mobile phase solvent blend used to perform the purification maintains the same ratio of weak (e.g. hexane) and strong (e.g. ethyl acetate) solvents throughout the chromatography. If you use thin-layer chromatography (TLC) as the basis for your flash chromatography, an isocratic purification would use the same solvent ratio as that used with the TLC.
With gradient elution the solvent blend at the end of the purification is different than at the beginning. Typically, gradient methods start with a mobile phase with a higher weak solvent percentage and ends with a higher strong solvent percentage.
There are actually two types of gradients, linear and step, and they have their own advantages that I’ll discuss later.
Isocratic Elution
As mentioned, isocratic flash chromatography uses a constant solvent blend to separate and elute a sample’s components. Isocratic elution is simple but does have some drawbacks, the greatest being band-broadening. As the crude sample migrates through the column, compounds with increasing affinity for the column media (stationary phase) elute more slowly than compounds with a greater affinity for the mobile phase. Since compounds migrate through the column as a concentration band, the longer it takes for a compound to elute from the column, the more solvent is required which increases the compound’s elution volume. Think of this as a dilution effect which is seen as elution band-broadening, Figure 1.
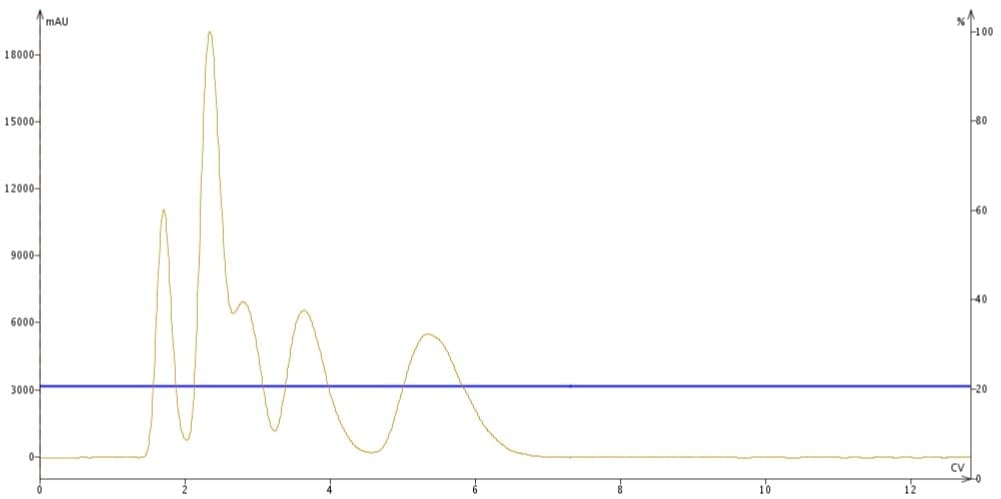
Figure 1. Isocratic flash chromatography. Compounds requiring longer elution volumes have broader elution bands seen here as peaks.
Gradient Elution
By changing mobile phase elution strength during the purification, later eluting compounds’ solubility in the mobile phase increases and expedites their elution. In doing so, these compounds’ elution bands sharpen, which increases their concentration and decreases band-broadening. As an added benefit, often the amount of separation between eluting compounds improves, thus providing higher purity fractions.
There are two types of gradients that are frequently used, linear and step. With linear gradients, the change in solvent ratio increases in a linear fashion expediting the elution of the compounds with greater stationary phase affinity and improving the separation, Figure 2.
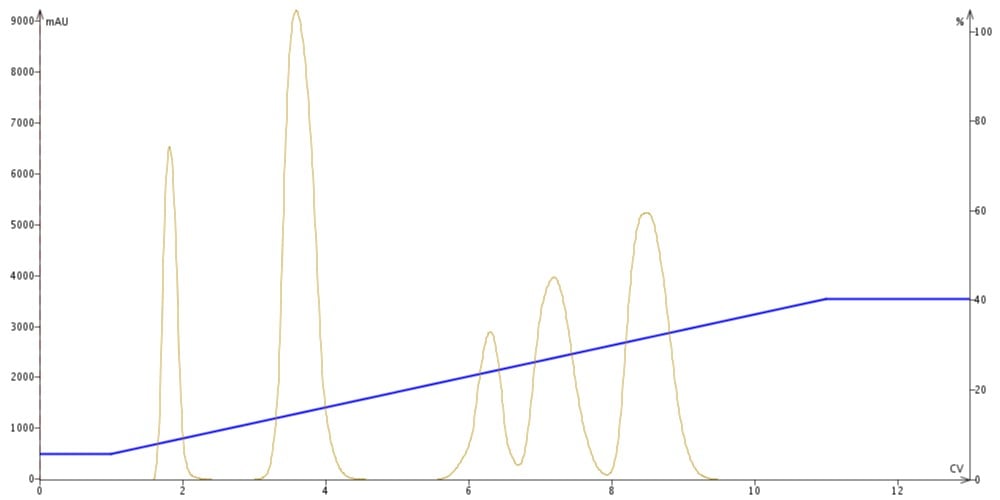
Figure 2. Linear gradient elution. As the elution solvent strength increases compound elution bands decrease improving purification efficiency.
A step gradient, on the other hand, is built with a series of isocratic steps (think stairs). Each consecutive step is a different solvent ratio containing a higher percentage of strong solvent, Figure 3. The idea behind step gradients is to control individual compound elution as much as possible in an effort to maximize the separation between compounds. Also, a frequent added benefit of step gradients is a reduction in the amount of solvent required to perform the purification.
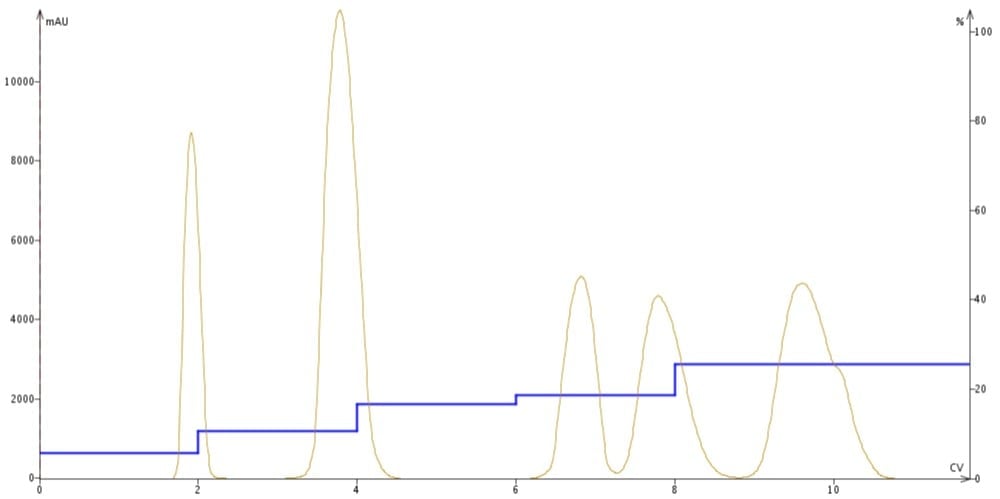
Figure 3. A series of individual steps helps to improve and maximize compound separation while minimizing band-broadening.
So, what we can see is that gradient elution, whether linear or step, improves flash chromatography efficiency and performance compared to isocratic elution.
Interested in learning more about flash chromatography? Download our whitepaper – successful Flash Chromatography

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership
