Media particle size and solvent flow rate play major roles in chromatographic separations including flash purification. This is true in both reversed-phase chromatography (aka partition chromatography) as well as normal-phase chromatography. The roles played are related to the overall compound mass-transfer kinetics and diffusion/dispersion as they migrate through the column. Smaller particles reduce sample dilution by reducing interstitial volume, while flow rate impacts the ability of molecules to efficiently pass through the porous particles. In this post, I will show how both particle size and flow rate impact flash chromatography.
A while ago I posted on this topic using reversed-phase columns. Since then I have been asked to address this issue with normal-phase silica columns to answer today’s question: How do particle size and flow rate affect normal-phase flash column chromatography? Because the separation mechanisms between normal- and reversed-phase are different (adsorption/desorption vs. partition), it really isn’t a bad idea to ask the question – do the variables particle size and flow rate play the same roles and have the same impact as in reversed-phase? The chromatographer in me says of course they do, but let’s test this hypothesis and show the data.
To generate these data, I created an isocratic method using 80% hexane and 20% ethyl acetate. My flash chromatography system is the brand new Biotage® Selekt, and to generate the separation, I used both 20 µm and 60 µm spherical silica packed in the new 5 gram Biotage® Sfär column design.
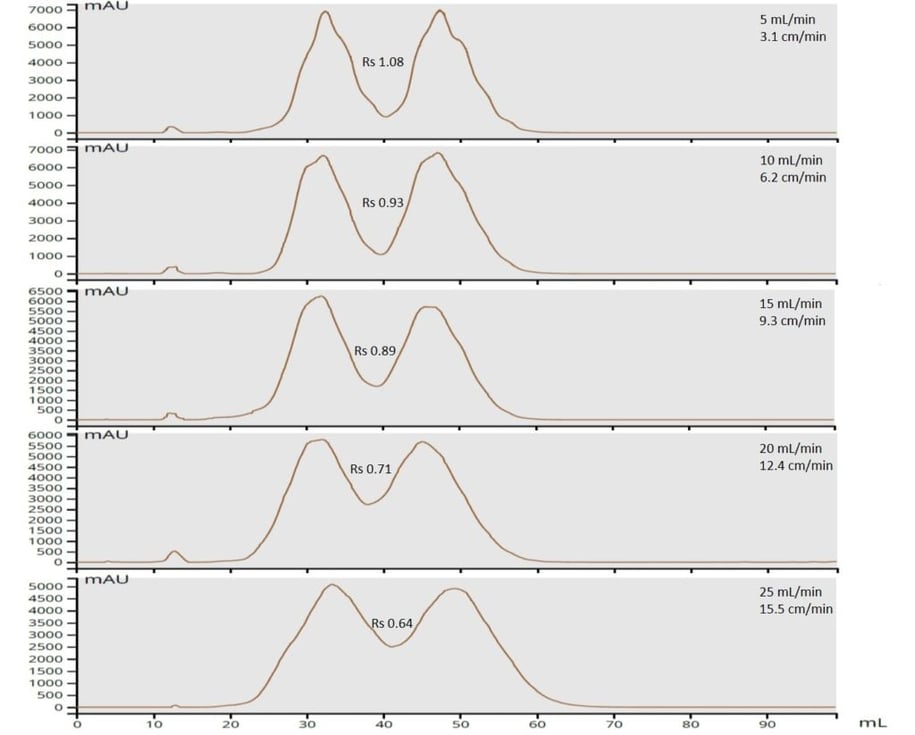
My sample was a 1:1 mixture of methyl (100 mg/mL) and butyl paraben (100 mg/mL) dissolved in acetone. Detection was UV photo-diode array (all wavelengths between 200 and 400 nm). To keep detector response on scale for the experiments, I kept my injection volume low (0.1 mL). Flow rates were changed between runs and included 5 mL/min, 10 mL/min, 15 mL/min, 20 mL/min, and 25 mL/min. Compound retention and peak width were measured by hand (anybody remember doing this?) and resolution calculated based on these values. Resolution impacts both load capacity and fraction purity, more resolution = more separation = higher purity/higher loading capacity, Table 1.
| Flow rate (mL/min) | 5 | 10 | 15 | 20 | 25 |
| Linear velocity (cm/min) | 3.1 | 6.2 | 9.3 | 12.4 | 15.5 |
| Resolution (20 µm silica) | 1.41 | 1.25 | 1.39 | 1.30 | 1.24 |
| Resolution (60 µm silica) | 1.1 | 0.9 | 0.9 | 0.71 | 0.64 |
The data shown in Figure 1 is a compilation of the chromatographic runs using the 60 µm media. From these data, you can clearly see that as flow rate (linear velocity) increases, the separation (resolution) between the parabens decreases from a high of 1.08 down to a low of 0.64 (or 41%).As I have previously posted, when it comes to solvent flow rates it is important to think (and scale-up) in terms of linear velocity in order to equate the flow between different column sizes.
However, with the smaller 20 um silica the resolution decrease is far less dramatic as flow increases, only decreasing from 1.41 to 1.24 (or 12%), Figure 2.
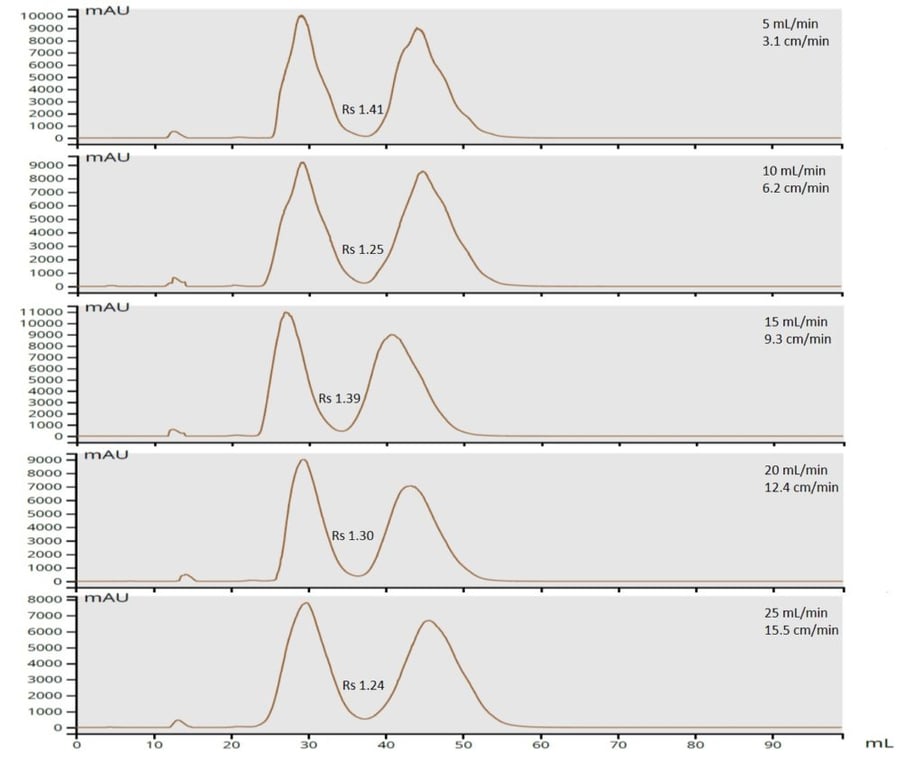
What these data ultimately show is that with smaller diameter particles you can achieve essentially the same separation regardless of flow velocity. This is quite important since the ability to increase flow rate (without much risk to fraction purity) reduces purification time and solvent consumption (more sample load per gram of silica with smaller particles). Data also show that with larger particle media, solvent flow velocity and sample load should be reduced in order to maximize compound resolution (load/purity).
So, when using flash purification and want to maximize throughput by increasing flow rate and sample load (while reducing solvent), you should consider using small particle, high surface area silica columns at elevated flow velocities.
To learn more about Sfär, follow the link here!

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership